August 8, 2016 — ClearFlow Inc. announced in late July that its PleuraFlow Active Clearance Technology is now available for the treatment of pediatric cardiothoracic surgery patients. After receiving clearance from the U.S. Food & Drug Administration (FDA) earlier this year for expanded Indications for Use with the company’s patented technology, ClearFlow developed a new model of the PleuraFlow product for the pediatric market.
All patients undergoing heart surgery have some degree of bleeding, and thus, all patients require chest tubes to evacuate shed blood from around the heart and lungs in the early hours after surgery. The problem is that chest tubes often clog, which can limit the ability to evacuate shed blood. This can lead to complications including retained blood around the heart and lungs.
The composite of drainage-related complications that are detrimental to outcomes after cardiothoracic surgery and that may require early or late intervention to remediate is known as Retained Blood Syndrome (RBS). These clinical complications have significant economic consequences, including higher costs of care for patients, hospitals and society at large.
Inadequate drainage due to chest tube clogging is especially a problem for smaller diameter tubes required for pediatric heart surgery. When intensive care unit (ICU) care providers are concerned with chest tube clogging, they often resort to makeshift bedside techniques to strip or milk out clots, or break the sterile field and advance suction catheters or balloon catheters into the tube in an attempt to re-open them after they have clogged. These techniques are ineffective and potentially harmful.
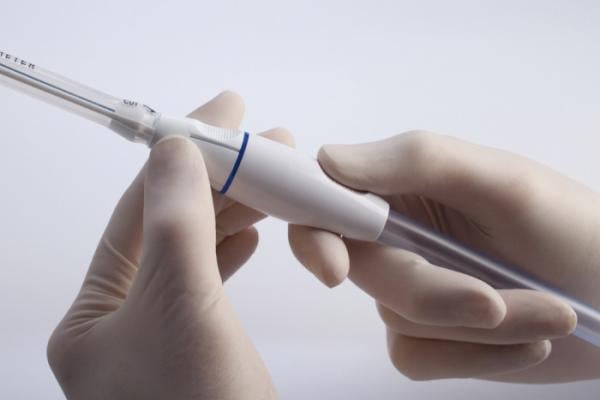
The PleuraFlow System is a medical device that removes clots from chest tubes. A clogged chest tube can lead to retained blood around the heart and lungs. The new, smaller sized options are designed specifically for pediatric applications.
“We commonly hear from pediatric heart surgeons, pediatric intensive care specialists and ICU nurses that they encounter problems with chest tube clogging after heart surgery in children, and sometimes this results in preventable major complications or even fatal consequences. The older makeshift bedside techniques of stripping or milking conventional chest tubes has been shown ineffective to prevent these problems, and can even be harmful, and thus are banned in many hospitals,” said ClearFlow CEO Paul Molloy.
In a study published in the Annals of Thoracic Surgery, smaller 20 Fr chest tubes fitted with Active Clearance Technology cleared blood significantly better than larger 32 Fr chest tubes without active clearance technology. This was the first time it was demonstrated that smaller tubes can drain better than larger tubes if the lumen is preventatively kept clear of clot. The FDA recently cleared a 20 Fr System with four side holes, which is ideally suited for infant, preadolescent and adolescent patients undergoing congenital cardiac surgery. This system is also available in a 20 Fr with six side holes, as well as 24 Fr System when needed for larger pediatric patients.
For more information: www.clearflow.com


 February 06, 2026
February 06, 2026