May 16, 2019 — Johnson & Johnson Medical Devices Companies announced the launch of Biosense Webster Inc.’s Cartonet to help electrophysiologists, hospitals, health systems, and researchers leverage and share data, with the goal of improving patient outcomes and operational efficiency in catheter ablation procedures. Announced at Heart Rhythm 2019, the Heart Rhythm Society's (HRS) 40th Annual Heart Rhythm Scientific Sessions, May 8-11 in San Francisco, Cartonet is the world’s first cloud-based networking and data analytics software solution for the field of electrophysiology, according to the company.
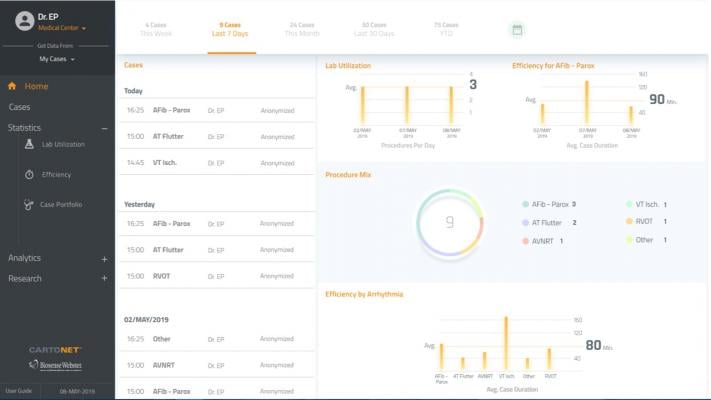
Cartonet gives doctors and hospitals the ability to review, analyze and share case data, along with video and image files obtained during catheter ablation procedures with Biosense Webster’s Carto 3 three-dimensional heart mapping system. With Cartonet, Carto 3 systems data will be stored on a secure cloud-based network where doctors can remotely access it via laptop, tablet or desktop. An interactive clinical dashboard provides statistics and analytics on case volume, lab utilization and procedural efficiency, among other data points. In addition, case data may be shared between institutions for clinical and research purposes.
Cartonet leverages the teamplay Cloud Platform of Siemens Healthineers, one of the largest data networks in healthcare with more than 3,600 connected institutions in 59 countries. The teamplay Cloud Platform meets industry best practices of security and privacy and supports compliance with HIPAA and GDPR.
“This is a very exciting development for the field of electrophysiology in that we will be able to more powerfully harness catheter ablation data to hone our own processes in the lab and to generate evidence and collaborate on research that could lead to new advances and improved patient outcomes,” said Jose Osorio, M.D., director, cardiac electrophysiology, Grandview Medical Center, Birmingham, Ala., who used Cartonet in a pilot program prior to its launch.
Catheter ablation is a proven safe and effective non-surgical treatment option for heart rhythm disorders including atrial fibrillation (AF). It works through neutralizing the abnormal electrical activity that causes an irregular heartbeat. Most patients who have the procedure experience a long-term reduction in the number of episodes of arrhythmia and the severity of symptoms. A high percentage achieve a permanent return to normal heart rhythm.
For more information: www.biosensewebster.com

 July 14, 2025
July 14, 2025