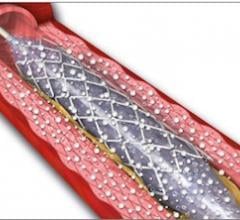
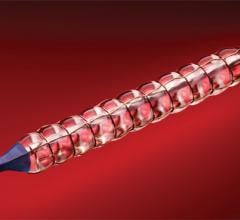
November 1, 2012 — It looks as though drug-eluting balloons (DEBs), already widely used in Europe, are set to burst into ...
Balloon Catheter
This channel includes news and new technology innovations for angioplasty balloon catheters (PTA). These are used in arteries with atherosclerotic lesions to compress the plaque expand the artery lumen to reopen occluded or heavily stenosed atherosclerotic lesions. Balloons are often used in combination with a stent to prop the treated vessel segment open. In addition to plain old balloon angioplasty (POBA), this section includes news about drug-coated balloon (DCB), valvuloplasty balloons and specialty cutting balloon.
October 17, 2012 — AngioDynamics entered into a definitive agreement to acquire all the outstanding capital stock of ...
September 28, 2012 — Boston Scientific Corp. announced it has received U.S. Food and Drug Administration (FDA) clearance ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
September 21, 2012 — Following the success of Biotronik’s Pulsar-18 0.018-inch, 4 French platform self-expanding stent ...
September 21, 2012 — Cardionovum GmbH announced that the results of a preclinical study and a first-in-man clinical ...
Austin Medical Design's Cath Cube is a single-use product for management of guidewires and catheters (balloons and ...
July 27, 2012 — C. R. Bard Inc. announced that its Lutonix technology center has completed patient enrollment into its ...
July 16, 2012 — TriReme Medical Inc. (TMI) announced that it has received U.S. Food and Drug Administration (FDA) ...
July 9, 2012 — r4 Vascular Inc. announced clearance from the U.S. Food and Drug Administration (FDA) to market the ...
July 9, 2012 — Miracor Medical Systems GmbH announced that the first three ST segment elevation myocardial infarction ...
July 9, 2012 — Cardionovum GmbH announced it has initiated in vivo testing of its second drug-eluting balloon (DEB) Rest ...
June 21, 2012 — TriReme Medical Inc. announced today it received U.S. Food and Drug Administration (FDA) clearance for ...
June 13, 2012 — Physicians presented at EuroPCR 2012 the results of two multicenter, randomized controlled trials, the ...
May 30, 2012 — Boston Scientific Corp. announces the U.S. Food and Drug Administration has cleared an expanded use ...
May 23, 2012 - TriReme Medical Inc. announced approval by the U.S. Food and Drug Administration (FDA) of the Glider PTCA ...

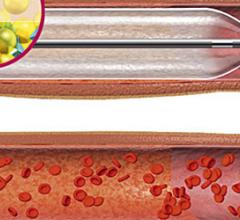
 November 01, 2012
November 01, 2012