Philips Healthcare launched the Epiq ultrasound system, a first-of-its-kind ultrasound…
Bayer HealthCare announced that the U.S. Food and Drug Administration (FDA) has cleared two new…
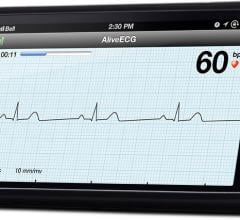
AliveCor Inc. released a new AliveECG App for its Heart Monitor that features an Enhanced Filter…
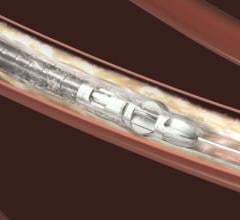
The U.S. Food and Drug Administration (FDA) approved Boston Scientific’s IntellaTip MiFi XP…
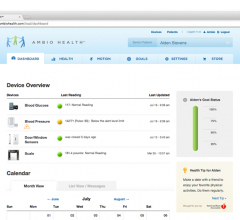
August 12, 2013 — Ambio Health, a healthcare technology company specializing in health…
NDS Surgical Imaging (NDSsi) has released its DomeAccess remote …
Sorin Group has received 510(k) clearance for commercialization of Sorin Connect, Sorin Group’s…
The U.S. Food and Drug Administration (FDA) recently granted market clearance for a hand lotion…
The Mikro-Cath disposable blood pressure catheter delivers accurate and reliable hemodynamic…
August 1, 2013 — Boston Scientific Corp. has received U.S. Food and Drug Administration (FDA)…
Polymer Technology Systems Inc. has introduced the CardioChek Plus analyzer. It is a combination…
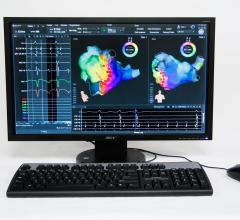
Nick Cavros, interventional cardiologist with Cardiovascular Institute of the South (CIS) at…
To assist providers in managing high-dollar inventory, McKesson has introduced radio frequency…
The Boston Scientific Corporation has received U.S. Food and Drug Administration (FDA) clearance…
Heart IT has released version 8 of its flagship products: WebPax …
IMRIS has announced U.S. Food and Drug Administration (FDA) 510K clearance to market VISIUS iCT…
Toshiba America Medical Systems Inc. has partnered with Unfors RaySafe Inc. to offer a new …
July 18, 2013 — Boston Scientific Corp. has received U.S. Food and Drug Administration (FDA) 510…
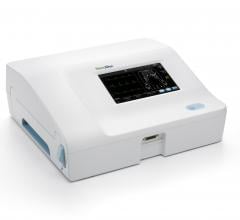
Welch Allyn announced the availability of two connectivity-ready electrocardiograph devices in…
July 15, 2013 — Leonhardt Ventures and Core Manufacturing LLC have announced a new partnership…


 August 30, 2013
August 30, 2013