April 27, 2010 – The U.S. Food and Drug Administration (FDA) this week cleared a large field-of-…
April 26, 2010 – A vascular closure device using a single suture fitted with stainless steel…
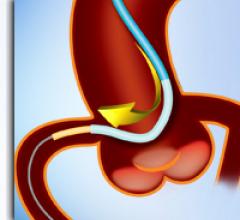
April 22, 2010 – Instead of using a conventional spring coil design, a new guidewire uses a…
April 22, 2010 - With the proliferation of telemedicine and tereradiology services, the two…
April 19, 2010 – A microvolt T-wave alternans (MTWA) original equipment manufacturer (OEM)…
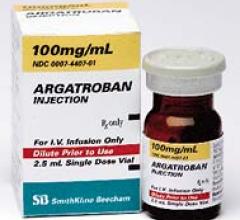
April 19, 2010 – The U.S. Food and Drug Administration (FDA) recently cleared a generic version…
April 14, 2010 – An antibacterial wrap for implanted pacemakers and defibrillators released this…
April 13, 2010 – The U.S. Food and Drug Administration (FDA) recently cleared a combined…
April 13, 2010 – A catheter designed to deliver embolic materials and radiopaque media to…
April 5, 2010 – The U.S. Food and Drug Administration (FDA) today said it approved the first…
April 2, 2010 – Integrating a sleep disorder diagnostic tool into hospital cardiac care, the…
April 1, 2010 – A new partnership will deliver multimodality cardiovascular image analysis as a…
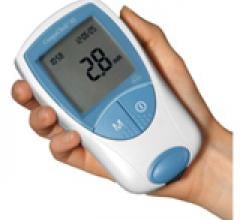
March 31. 2010 – The FDA granted Clinical Laboratory Improvement Amendments (CLIA)-waived status…
March 30, 2010 – The FDA granted tentative approval for Teva Pharmaceutical’s abbreviated new…
March 29, 2010 – A new multiparameter ambulatory monitoring solution that transmits…
March 26, 2010 – The FDA granted 510(k) market clearance for the AngioSculpt PTA Scoring Balloon…
The GuideLiner catheter is a coaxial “mother and child” guide extension using a rapid exchange…
A new and improved design of the original Gopher catheter, the Gopher Gold is designed for use…
The Merit Laureate hydrophilic guide wire is designed for drainage catheter access, dialysis…
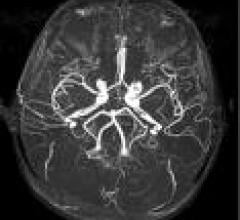
March 23, 2010 - When imaging the brain, time is critical as vascular abnormalities can have a…


 April 27, 2010
April 27, 2010