The tricuspid valve has been dubbed “the forgotten valve” by many practitioners in the interventional cardiology space due to a lack of transcatheter repair and replacement devices, unlike the aortic and mitral valves. Early developments focused on the other two valves because they were easier to visualize and reach. As TAVR becomes a mature technology and numerous companies are in the middle of development or trials for transcatheter mitral devices, recent R&D efforts by vendors have shifted to tricuspid devices. The promise shown in these devices in early trials is generating great enthusiasm. Clinical trial data for these devices were presented in June at the 2018 Transcatheter Vascular Therapies (TVT) annual meeting in Chicago.
“It’s an exciting time because the technologies are ripe to be implemented on the tricuspid valve,” said Ori Ben-Yehuda, M.D., executive director of the Cardiovascular Research Foundation’s (CRF) Clinical Trials Center. “We’ve learned a lot from using these same technologies on the right side, and we have a very large patient population who are in need of these therapies.”
Challenges of the Tricuspid Valve
Tricuspid valve insufficiency, also known as tricuspid regurgitation (TR), affects an estimated 65-85 percent of the population,[1] with the true prevalence of moderate to severe TR estimated at 1.6 million people in the United States.[2]
There are several elements that make the tricuspid valve a more difficult proposition for treatment, beginning with the anatomy of the right side of the heart.
“The difficulty in this field is we don’t know as much about the tricuspid valve as the mitral valve, and the anatomy of the tricuspid is so different,” said Rebecca Hahn, M.D., professor of medicine and director of interventional echocardiography at Columbia University Medical Center in New York. As an example, Hahn noted that the tricuspid valve does not have a set number of papillary muscles. “There’s usually one standard single papillary muscle, but there may be a posterior papillary muscle, maybe five or six septal papillary muscles, so it’s very variable. And that high amount of variability makes it difficult to understand this valve.”
Because of these anatomical differences, Ben-Yehuda said, the devices and procedures that work on left-sided heart disease will not necessarily work on the right side of the heart. “Even assessing right ventricular function, which is so central to assessing tricuspid disease, is not a well-developed methodology compared to assessing the left ventricle. The right ventricle has a different geometry, it’s much more challenging,” he said. “So as these therapies are being developed, we’re also developing our understanding of the underlying disease.”
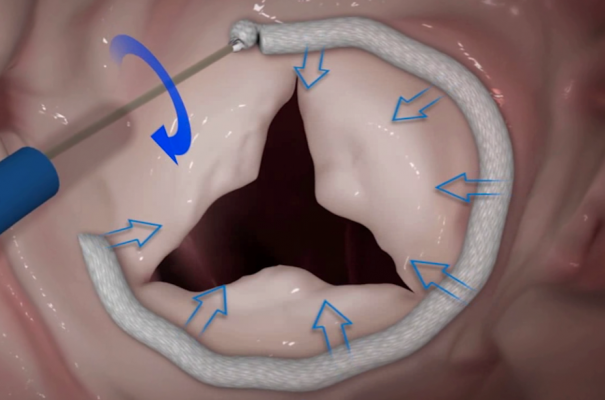
Edwards Cardioband Gains Approval in Europe
At present, the only transcatheter tricuspid device approved for patient use anywhere in world is the
Edwards Cardioband Tricuspid Valve Reconstruction System, which received the European CE mark approval in May. The device is delivered via a transfemoral approach and is designed to reduce tricuspid regurgitation through annular reduction. It enables precise positioning to a patient’s specific anatomy and real-time adjustment, with simultaneous confirmation of results. The Cardioband Tricuspid System follows the same design and implant technique as its predecessor, the Cardioband Mitral System.
The Cardioband Tricuspid System received European approval based on results from the TRI-REPAIR Study, a single-arm, multi-center, prospective study in Europe evaluating device performance and safety. The primary safety endpoint was overall rate of major serious adverse events (MSAEs) and serious adverse device effects (SADE) until hospital discharge and at 30 days post-procedure. Technical success measures included:
● Successful access, deployment and positioning of the Cardioband device; and
● Septolateral reduction at intra-procedure and discharge.
Results were reported at the 2018 Transcatheter Vascular Therapies (TVT) meeting in June by Prof. Georg Nickenig, chief, Department of Cardiology, University Hospital, Bonn, Germany. All 30 patients experienced successful access, deployment and positioning of the Cardioband Tricuspid device. Patients saw an average 16 percent reduction in septolateral diameter, and annular reduction was sustained at six months.[3]
Trialign Device Reduces TR Following Retreatment
● A 21 percent improvement in six-minute walk test;
● A 21.6 percent reduction in PISA EROA (a measure of regurgitation) from baseline; and
● A 17.1 percent improvement in left ventricular outflow tract (LVOT) stroke volume from baseline.
“The SCOUT I study is the first, Early Feasibility Study in the U.S with core lab adjudicated one-year data showing long-term improvement in a population of subjects with severe, progressive TR,” Hahn stated at TCT. “Patients in the study continue to report feeling better, which is supported by sustained improvement in multiple clinical and quality of life measures.”
A two-year case was presented recently at TVT 2018. The patient was a 76-year-old female in NYHA Class III who presented with severe-massive tricuspid regurgitation. She had previously had mitral valve surgery and was enrolled in SCOUT I. The treatment had been deemed successful, and the TR was reduced to moderate-mild using just one pair of pledgets. At year two, however, the patient became symptomatic. She was re-treated with Trialign and two more pairs of pledgets were added. The re-treatment reduced PISA by 50 percent, and 30-day results showed mild TR with the patient reporting improved quality of life and clinical outcomes.[4]
Enrollment Begins in TRILUMINATE Trial
While annuloplasty systems offer one promising possibility for treating tricuspid regurgitation, Abbott is taking a different approach with its clip-based transcatheter tricuspid valve repair (TTVR) system currently enrolling in clinical trials.
The company launched the TRILUMINATE trial in August 2017, with the first patient enrolled at the Minnesota Heart Institute Foundation the following month.
Abbott’s TTVR system builds upon the company’s MitraClip System to treat mitral regurgitation. In the mitral valve, the system secures a portion of the leaflets with an implanted clip to prevent the backflow of blood.
The TRILUMINATE study is a prospective, single-arm, multi-center study designed to evaluate the performance of clip-based technology in approximately 75 asymptomatic patients at 25 sites across the United States and Europe. The primary endpoints are an echocardiographic tricuspid regurgitation reduction of more than or equal to 1 grade at 30 days post-procedure, and the assessment of major adverse events at six months.
"Current pharmacological and surgical treatment options are not meeting the needs of people living with tricuspid regurgitation," said Nickenig, who is the lead investigator of the study. "Abbott's MitraClip has shown positive results for mitral regurgitation, and we hope this study shows that a similar clip-based technology may effectively treat people with tricuspid regurgitation."
Ben-Yehuda noted that real-world results suggest promising outcomes for the TTVR system. “The MitraClip, in terms of actual use in a registry setting, has had the most experience in terms of treating patients with TR using a transcatheter approach,” he said.
Related Transcatheter Tricuspid Valve Technology Content:
References:
3. Nickenig G. Cardioband Tricuspid Showing Promise Technology and Clinical Updates. Presented at TVT annual meeting, June 21, 2018.
4. Meduri C.U. Severe TR Retreated Two Years Post Initial Procedure. Presented at TVT annual meeting, June 21, 2018.


 November 14, 2025
November 14, 2025 









