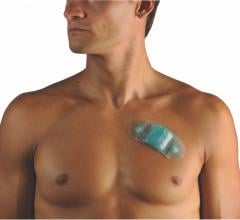
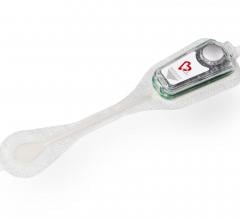
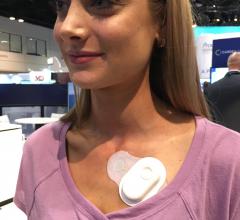
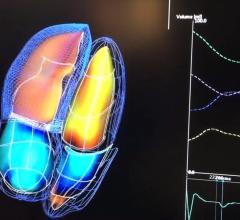
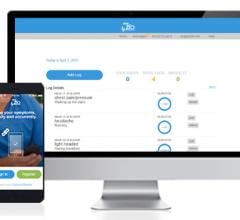
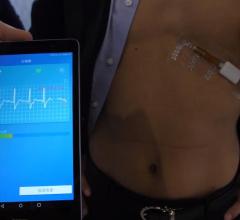
May 23, 2018 — Medtronic announced on May 23, 2018 that it will discontinue marketing for its SEEQ Mobile Cardiac ...
Wearables
This channel includes news and new technology innovations for wearable cardiac diagnostic systems. These include technologies related to smart phone apps, wearable monitors, consumer-grade wearables and medical grade FDA cleared wearables. A good overview on the future of these technologies can be found in this article.
May 17, 2018 — A new study published in JAMA Cardiology used the Zio continuous cardiac monitoring system by iRhythm to ...
May 16, 2018 — A new study is the first to validate the accuracy of wrist-worn wearable devices in measuring induced ...
Technology-enabled “Insourcing” approach to cardiac arrhythmia diagnosis provides patient and provider benefits The ...
May 8, 2018 — Bardy Diagnostics Inc. (BardyDx) announced that the American Heart Journal has published the results of a ...
I have been looking for new ways to show readers interesting things I find during the medical conferences I attend, so ...
The show floors at the American College of Cardiology (ACC) 2018 meeting in March and at the Heart Rhythm Society ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
DAIC Editor Dave Fornell takes a tour of some of the most interesting new technologies on the expo floor at the 2018 ...
March 19, 2018 — For people at heightened risk for atrial fibrillation (AFib), a common heart rhythm disorder that also ...
March 15, 2018 – A newly-designed wristband and corresponding app that works with a smartwatch can accurately display ...
COVID-19 has posed challenges for physicians whose cardiac patients are at-risk and reluctant to schedule an office ...
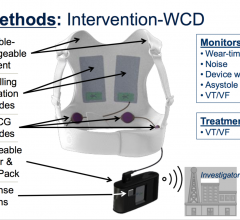
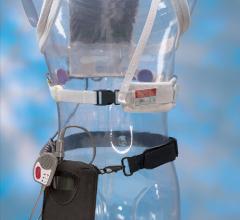
March 13, 2018 – Wearing a lightweight vest equipped with a cardioverter defibrillator that detects abnormal heart ...
Take a video tour of some of the medical devices designed to improve patient care, improve patient engagement and ...
January 22, 2018 — The U.S. Food and Drug Administration (FDA) announced last week it is providing information and ...
COVID-19 has posed challenges for physicians whose cardiac patients are at-risk and reluctant to schedule an office ...
December 28, 2017 — Zoll Medical Corp. and Myant, Inc., a high-tech advanced manufacturing company, announced they have ...
December 12, 2017 — VitalConnect Inc. announced its fifth 510(k) market clearance from the U.S. Food and Drug ...
November 3, 2017 — VitalConnect Inc. announced the fourth and final Series C closing, bringing the total Series C round ...


 May 23, 2018
May 23, 2018