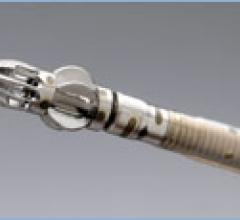
Vascular Solutions' Jiffy Wire, an 80 cm 0.035-inch specialty guide wire, was launched in July…
St. Jude Medical said the FDA cleared its SJM Confirm implantable cardiac monitor (ICM), a…
The ClariTEE miniaturized transesophageal echocardiography probe facilitates episodic monitoring…
March 23, 2009 - Pathway Medical Technologies Inc. today said the FDA granted the company 510(k…
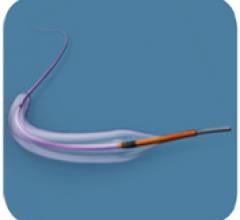
January 31, 2011 – A new catheter used to treat coronary artery disease has received regulatory…
The U.S. Food and Drug Administration (FDA) today expanded the indicated use for the CoreValve…
The LATITUDE Patient Management System by Boston Scientific has FDA approval to be used in…
Calgary Scientific Inc. Medical Group (CSIMG) announced the receipt of FDA 510(k) clearance for…
Boston Scientific Corp.
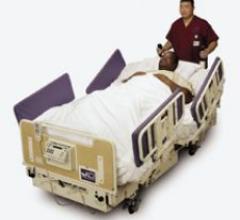
The BariMaxx II Power Drive System is a 1,000-pound capacity expandable side exit bariatric bed…
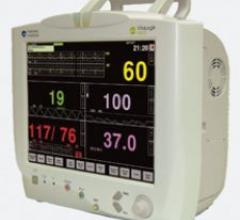
Monitoring system maker Mennen Medical recently said an additional monitor, the VitaLogik 4000…
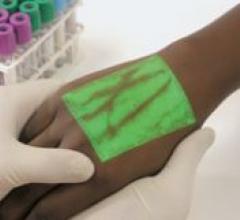
The VeinViewer by Luminetx is a vascular imaging system that allows physicians, nurses and other…
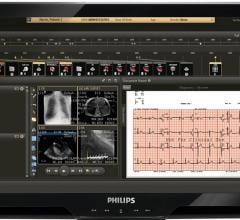
TomTec Imaging Systems recently announced that its new CardioArena, the network and imaging…
Stereotaxis’s suite of proprietary magnetic coronary guidewires including CRONUS, ASSERT and…
The FDA has cleared Medtronic’s Attain StarFix OTW (over-the-wire) lead (Model 4195), an active…
he Powerheart AED G3 is designed with Rescue Ready technology that self-checks all main…
Vascular Solutions Inc. offers the EXPRO Elite Snare, a one-piece medical device designed for…
Mennen Medical recently released a new cath lab hemodynamic monitoring system, the Horizon XVu,…
April 6, 2009 – At HIMSS 2009, KeyScan showcased its new keyboard scanner that is upgraded to…


 October 19, 2008
October 19, 2008