December 30, 2022 — Azmi Atiya, MD, an established and highly respected cardiothoracic surgeon in the San Fernando ...
Endovascular Aortic Repair
Endovascular aortic repair (EVAR) in a transcatheter method to repair abdominal aortic aneurysms (AAA) and thoracic endovascular aortic aneurysm repair (TEVAR). The procedure usually involves placement of self-expanding,covered stent grafts to close off the aneurysm and prevent further aortic leaks. This can be used instead of open vascular surgery. EVAR today accounts for the majority of aortic aneurysm repairs compared to surgery.
December 14, 2022 — Edwards Lifesciences identified the top data releases from 2022 that contributed most to shaping ...
December 5, 2022 — Imagen Technologies, Inc. announced the U.S. Food and Drug Administration's 510(k) clearance of the ...
November 2, 2022 — The second round of late-breaking clinical trial results were announced at VIVA22 on Nov. 1 in Las ...
November 2, 2022 — A number of awards of distinction were presented during The VEINS (Venous Endovascular INterventional ...
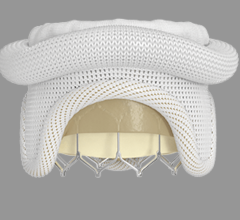
October 26, 2022 — Thousands of people have new hope for treatment of thoracic aortic arch disease and University ...
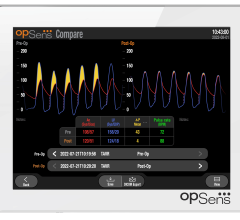
October 6, 2022 – OpSens Inc., a Quebec-based medical device cardiology-focused company, has announced that it has ...

The 34th Transcatheter Cardiovascular Therapeutics (TCT), the annual scientific symposium of the Cardiovascular Research ...
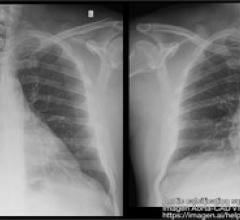
September 13, 2022 — The ŌNŌ endovascular retrieval system was recently used to remove a malpositioned leadless ...
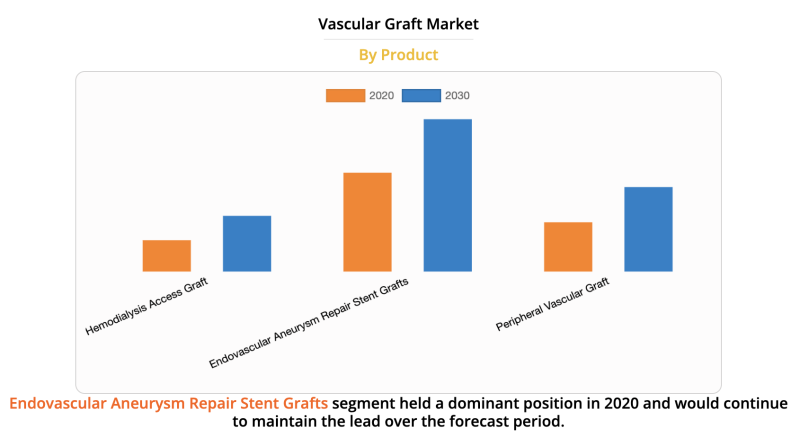
The global vascular graft market size was valued at $4,993.64 million in 2020, and is projected to reach $8,138.68 ...
June 14, 2022 — A new American Heart Association scientific statement highlights the need for more data and research ...
May 25, 2022 — ŌNŌCOR LLC, a leader in endovascular safety technology, today announced it has received 510(k) U.S. Food ...
May 19, 2022 — One year outcomes from the Disrupt PAD III Trial comparing intravascular lithotripsy (IVL) with a drug ...
May 19, 2022 — Cardionomic, Inc., a Minneapolis medical device company, is pleased to announce initial enrollment in ...
May 4, 2022 – Seeking to bolster its development of a biocompatible graft that promises to reshape the future of cardiac ...


 December 30, 2022
December 30, 2022