January 19, 2023 — Shockwave Medical, Inc., a pioneer in the development of Intravascular Lithotripsy (IVL) to treat ...
Cath Lab
The catheterization lab channel includes content related to interventional technologies for coronary and peripheral artery disease (PAD). Other cath lab transcatheter device technologies covered on this page included percutaneous treatment for stroke, venous interventions, heart valves, hypertension, heart failure and percutenous coronary interventions (PCI).
Behavioral science is being applied in unique ways for enterprise-imaging product design.
Behavioral science is the ...
Adjustable parameters on the ACIST® CVI® Contrast Delivery System include, flow rate, volume, pressure limit, rise time ...
January 12, 2023 — Selution SLR, MedAlliance’s novel sirolimus-eluting balloon, has received conditional FDA ...
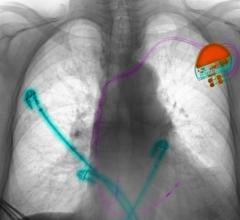
January 12, 2023 — Medtronic announced the first patient enrollment in the ADVANCE Trial, a head-to-head randomized ...
January 11, 2023 — The Alameda, Calif.-based global healthcare company Penumbra has announced U.S. Food & Drug ...
As medical advancements continue to push the boundaries of what is possible in the field of structural heart ...
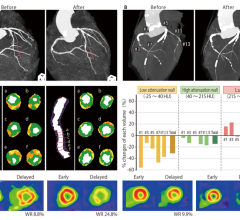
January 11, 2023 — In recent years, transcatheter mitral valve replacement (TMVR) treatment and technology has evolved ...
January 9, 2023 — ReCor Medical, Inc. and its parent company, Otsuka Medical Devices Co., Ltd. announced the appointment ...
January 9, 2023 — When we were little, our parents told us to take our vitamins so we could grow big and strong. Now ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
January 3, 2023 — The market has been studied for the below mentioned-segmentation and regional analysis for North ...

As 2022 comes to a close, the Diagnostic and Interventional Cardiology team takes a look at the year's most viewed ...
January 2, 2023 — The U.S. Food and Drug Administration (FDA) has approved the Stellarex 0.035” OTW Drug-coated ...
During cardiology fellowship, my institution had a hand manifold system. I found it difficult to use at times, and it ...
January 2, 2023 — The U.S. Food and Drug Administration (FDA) has approved the BioFreedom Drug Coated Coronary Stent (DC ...
The DAIC team wishes you a very happy and safe New Year!
December 23, 2022 — According to Coherent Market Insights, the global Drug Eluting Stents market is estimated to be ...


 January 19, 2023
January 19, 2023