May 26, 2009 – Medtronic intends to seek FDA approval for the use of cardiac resynchronization therapy (CRT) in ...
EP Lab
This channel includes news and new technology innovations for cardiac electrophysiology (EP) systems, techniques and devices using in EP labs. This includes implantable EP devices, pacemakers, implantable cardioverter defibrillators (ICD), cardiac resychronization therapy (CRT), ablation technologies, left atrial appendage (LAA) occlusion, atrial fibrilation (AF) and Holter monitors.
May 22, 2009 - According to IMV’s recent census of cath labs in the U.S., starting in 2007 the total number of ...
May 22, 2009 – Biotronik said today it is partnering with Belgian biotechnology company Cardio3 BioSciences in its ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
May 19, 2009 - MVP (Managed Ventricular Pacing), an exclusive programming feature on Medtronic pacemakers that has ...
May 18, 2009 – Stereotaxis Inc. displayed updated versions of its Odyssey Workstation, Cinema and Connect systems ...
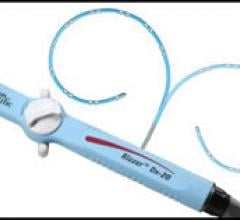
May 18, 2009 – Boston Scientific showcased its new Blazer Dx-20, a bidirectional duodecapolar diagnostic catheter ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
May 18, 2009 - With electrophysiology procedures continuing to grow in a time of increasing economic constraints ...
May 18, 2009 - CardioNet Inc. launched SomNet, a new clinical indicator for severe sleep disorders available in ...
May 18. 2009 - TYRX Inc. announced the initial, interim clinical results from its AIGISRX Antibacterial Envelope ...
Washington Health System (WHS) provides healthcare services at more than 40 offsite locations across three counties in ...
May 18, 2009 – At Heart Rhythm 2009 last week Biotronik said it completed the first enrollment in the pivotal ...
May 17, 2009 – GE Healthcare announced new and expanded collaborations with Hansen Medical Inc., STERIS Corp. and ...
May 14, 2009 - St. Jude Medical Inc. yesterday announced European CE mark approval of its Accent RF pacemaker and ...
Apple created a stir when it announced in 2018 that its Apple Watch Series 4 was the first consumer health and fitness ...
May 14, 2009 - Partnering with Healthworks Inc., the Philips Learning Center is offering a series of live lecture ...
May 14, 2009 – In final, one-year results of a multicenter randomized clinical study, catheter ablation ...
May 13, 2009 - Medtronic Inc. today announced the start of a nationwide trial to examine the relationship between ...

 May 26, 2009
May 26, 2009