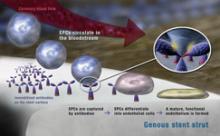
February 21, 2011 – Two drug-eluting coronary stent systems have been launched in India. Boston Scientific’s Promus Element everolimus-eluting stent system and Taxus Element paclitaxel-eluting coronary stent system incorporate the same novel platinum chromium (PtCr) alloy.
They also have the same stent design and advanced catheter delivery system.