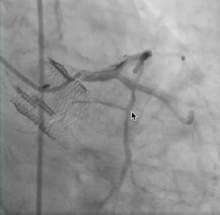
Orchestra BioMed Inc. has secured Breakthrough Device designation by the U.S. Food and Drug Administration (FDA) for its Virtue Sirolimus-Eluting Balloon for the treatment of coronary in-stent restenosis (ISR).
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now