The American College of Cardiology (ACC) annual meeting is a key event where vendors launch new cardiology technologies. After walking the entire expo floor at ACC.14, these are my choices for some of the most innovative new technologies that were on display.
At the Healthcare Information and Management Systems Society (HIMSS) annual meeting in February, one of the takeaways was that IT departments need to be prepared for a major influx of data in the coming years from an explosion of small, inexpensive patient monitoring devices. These devices will be used for both patient engagement efforts required under Stage 2 Meaningful Use and to better monitor patient health during the long periods between office visits. This trend was very evident on the show floor at ACC.14. Another trend seen at both HIMSS and ACC this year is the rapid expansion of mobile device apps for physicians to improve their workflow using smartphones or tablet devices.
Click to view the video version of this article with clips from the ACC show floor.
Implantable Cardiac Monitor
Earlier this year, Medtronic gained U.S. Food and Drug Administration (FDA) approval for its Reveal Linq implantable cardiac monitor (ICM) device. It replaces the first generation Reveal system, which was about the size of a USB thumb drive. In contrast, the Linq is 87 percent smaller, has 20 percent more memory and a three-year battery life. It also transmits patient data wirelessly. The device eliminates the need for bulky Holter or event monitors and their associated leads, especially in patients with extremely transient arrhythmic events.
The monitor’s small size also makes it much easier to implant. It only requires an incision about a centimeter wide, made with a pointed scalpel pushed into the patient’s skin, which is supplied with the implant kit. The device is loaded into an insertion device that creates a small pocket for the Linq under the skin and guides the device into the pocket using a syringelike plunger. An implant can be completed in a few minutes.
Simple-to-Use, Inexpensive Holter Monitoring
Traditional Holter monitors are expensive, require precise placement of several wire leads and are viewed as complicated to use by some patients and clinicians. To expand Holter use to non-expert clinical personnel, several companies have developed small, lightweight and simple-to-use devices.
iRhythm introduced the innovative Zio Patch a few years ago, which incorporates the Holter into a device about the size of a small pedometer with an adhesive backing that the patient sticks to their chest, eliminating the need for numerous wire leads, precise lead placement or the need for the patient to carry a belt-mounted box. At ACC.14, iRhythm was joined by other vendors that recently received FDA clearance for similar monitoring devices.
DMS Service was among these vendors, offering the MyPatch Holter device, which sticks to the patient’s sternum. The tiny Holter, weighing less than an ounce, is reused, but the adhesive lead pad it attaches to is disposable. Like the Zio, MyPatch is streamlined to eliminate most buttons to greatly simplify operation for both the clinical staffand the patient to improve compliance.
Last year Preventice gained FDA clearance for its BodyGuardian event or continuous monitoring system. It can monitor patients for up to 30 days and uses an extremely small, simplistic and inexpensive design to help reduce healthcare costs. The device is half the size of a smartphone, is rechargeable and uses disposable adhesive pads to attach directly to a patient’s skin. It comes with a smartphone that is used solely as a device monitor, receiving data wirelessly from the device. The phone displays battery life, heart rate, breathing rate, activity level and if proper lead contact is maintained. Waveforms can be wirelessly sent to a physician’s office for further review after an event is recorded.
Realizing the need for more simplistic, lightweight and mobile friendly Holters, GE Healthcare developed the
Seer 1000 Holter, launched at ACC.14. It has one-button operation and is about half the size of a smartphone. It uses a Bluetooth interface with a tablet device to exchange data. The system still uses traditional wire leads, but its tablet app shows pictures of where the leads need to be placed on the body and indicates if a lead is incorrectly placed and how to fix it. The app also shows battery life and dynamic waveforms for each lead. The simplicity of the device is hoped to expand Holter use by non-electrophysiology staffand at rural clinics.
Mobile ECG Review
A couple years ago, Airstrip was the first to introduce mobile access to dynamic ECG waveforms and ECG management systems via mobile phones and tablet devices. As medicine continues to become more mobile friendly, other vendors are now following suit. McKesson demonstrated its FDA 510(k)-pending McKesson ECG Mobile app, which offers a bi-directional link to McKesson’s ECG management system. It also offers a dynamic display of ECGs, and can call up prior ECGs to conduct overlays of the waveforms. It also has measuring calipers. The system was designed to use a minimal number of hand movements to improve workflow.
Improved ECG Management System Connectivity
GE Healthcare’s Muse has been a popular ECG management system in the U.S. market for years. However, users’ biggest complaint about Muse is its proprietary software, which has prevented or made difficult integration with non-GE ECG systems. In an era of increasing interoperability, GE took this criticism to heart and redesigned Muse to allow plug-and-play, bi-directional integration with most ECG systems on the market. Through its new i2 ECG Connect interface, Muse now enables integration of ECG data from devices manufactured by Philips, Mortar, Nihon Kohden, Edan and Schiller.
ECG Patient Engagement
Stage 2 Meaningful Use requires bi-directional patient engagement so patients will become more involved in their own health. An example of how this might work in cardiology is the use of the AliveCor single-lead ECG system, which converts any Android or iPhone device into an ECG recorder. The device was approved by the FDA in 2012 and generated a lot of interest on the show floor at ACC.13, but it was limited by the FDA indication requiring a prescription and there was no way for patients to evaluate ECGs on their own. In early 2014, AliveCor launched a Web-based ECG review service where patients can send an ECG from their phone for immediate review by a doctor or an ECG tech for a fee. The device also gained FDA clearance for over-the-counter sales to anyone who wants one. That combination has made for rapid expanding use of the device.
The app stores recorded ECG strips and notes, such as if the patient had symptoms, had performed physical activity or drank a cup of coffee. These can be e-mailed to a primary care physician or a cardiologist to include in the patient’s electronic medical record (EMR), or for immediate review during a cardiac event.
In 2013, AliveCor gave away 1,000 of these devices to patients with atrial fibrillation at a patient conference in the United Kingdom. A year later, the vendor says it is still receiving up to 700 ECGs a day from those patients, showing a very high level of bi-directional engagement.
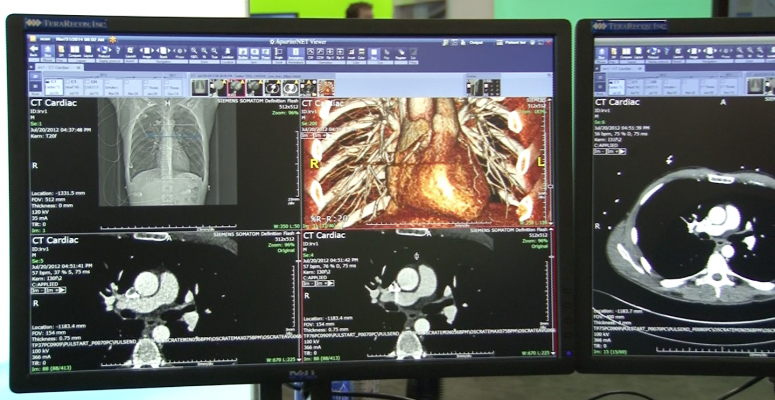
Less Expensive Options for Enterprise Imaging
There is a trend in medicine away from silos of data and images for each department toward enterprise-wide imaging and data access. This can be accomplished through use of a VNA or remote viewing software overlays, but a new and less expensive option was shown at ACC.14 by TeraRecon. Its iNteract Plus software acts as an integration tool, bridging various PACS and reporting systems, regardless of vendor or department. It can integrate images, waveforms, labs, reports and other patient data from various systems using links within a patient’s EMR. The system also can be used as a remote image viewing system using an existing PACS.
Fusing IVUS With Angiography
An issue with intravascular ultrasound (IVUS) in the cath lab is the guesswork involved when trying to figure out exactly where the IVUS is imaging inside a vessel on the angiographic imaging used for procedural navigation. At ACC.14, Volcano launched its new SyncVision IVUS/Angiography co-registration software. It enables playback of an IVUS pullback, which shows the precise location of the imaging tip of the IVUS catheter on a frozen angiographic view of the vessel. The co-registration can help operators more quickly and accurately identify anatomic landmarks, such as side branch vessels or specific lesions to be treated in vessels with long diffuse disease.






 February 09, 2026
February 09, 2026