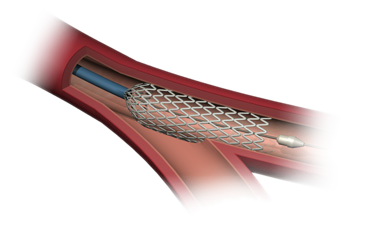
July 9, 2009 –Stentys said today it received CE mark approval for the European sale of its disconnectable and self-expanding stent platform for treatment of coronary artery bifurcations.
The stent is designed for treatment of acute myocardial infarction (AMI) and coronary artery bifurcations. The company also announced that six-month follow-up data from its OPEN I clinical study demonstrated safety and efficacy of its disconnectable and self-expanding platform in treating coronary lesions close to a bifurcation.
The versatile Stentys platform has two features - self-expansion and disconnectability. The self-expanding feature ensures optimal apposition in the critical initial hours and days after an AMI procedure by being constantly applied to the vessel surface during thrombus and vessel spasm relief, therefore avoiding malapposition, the company said. The disconnectability feature is designed to treat lesions close to a bifurcation, by ensuring safe main branch provisional stenting and optimal side branch access when needed. The company said the Stentys platform is implanted by usual stenting techniques, so it does not require additional training.
While the CE mark approval for bifurcations represents a very significant milestone for the company, it said it wants to demonstrate that AMI patients may be better treated with the self-expanding device.
For more information: www.stentys.com

