FFR-CT imaging developed by HeartFlow offers coronary flow data noninvasively. Clinical data so far shows close correlation with invasive catheter-based FFR measurements. The technology helps pinpoint ischemic culprit lesions and determine the severity of coronary stenosis.
Trends and new technology for cardiac computed tomography angiography (CCTA) were highlighted during the 2012 Society of Cardiovascular CT (SCCT) annual scientific meeting in July in Baltimore, Md. Major topics included the development of fractional flow reserve (FFR)-CT imaging, perfusion imaging, imaging for transcatheter aortic valve replacement (TAVR) and analysis software advancements.
FFR-CT
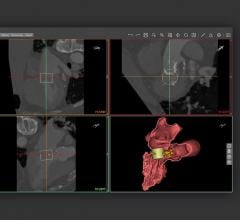
One of the exciting, next-step technologies for CCTA is FFR-CT developed by HeartFlow. This automated software can quantify the blood flow in each coronary segment, displaying it in a color-coded map on a 3-D coronary tree. If validated, the software may drastically reduce the need for diagnostic catheter angiography procedures. It also promises to pinpoint the specific culprit lesions in patients with myocardial ischemia.
FFR has become a popular tool in interventional cardiology to measure the impact of a lesion on coronary blood flow. Although a lesion may look severe on CT or during invasive angiography, clinical research[1, 2] has found that the hemodynamic flow might not be significantly impacted. Stenting hemodynamically insignificant lesions does not impact patient outcomes, decreases future revascularization options of that lesion, and significantly increases healthcare costs. Using a catheter wire to measure flow before and after a lesion in a vessel, a cardiologist can compute an FFR number. If 0.8 or higher, the lesion does not significantly impact flow. If less than 0.8, the flow is considered restricted and revascularization is required. Use of FFR has helped target only lesions causing ischemia, reducing the number of stents implanted.
FFR-CT looks at the flow of iodine contrast through a lesion. It is an automated program that uses complex computational fluid dynamics algorithms to estimate FFR. In comparisons between FFR-CT and actual FFR catheter measurements in the DISCOVER-FLOW trial, the technology appears to be very accurate. Additional positive validation data is expected from the DeFACTO trial, being presented at the European Society of Cardiology (ESC) meeting in August.
CT is very good at imaging anatomy and can visualize large plaques in the coronary arteries. However, a large plaque does not necessarily mean it is the cause of chest pain or ischemia, said James Min, M.D., director of cardiac imaging research and co-director of cardiac imaging, Cedars-Sinai Heart Institute, Los Angeles. He said that often patients with large lesions detected on CT are sent to the cath lab, where it is found the lesions are hemodynamically insignificant based on FFR.
“What FFR-CT is really doing is increasing the specificity of CT in imaging lesions, increasing accuracy by 25 percent,” Min said. “The potential is to identity the lesions that cause ischemia.”
An advantage of FFR-CT, beyond being noninvasive, is that adenosine does not have to be administrated, as is required with invasive FFR measurements. FFR-CT exams do require the administration of nitroglycerin prior to the scan to dilate the coronary vessels for better image quality.
Currently, the software is not available on hospital workstations because it requires so much computing power. Instead, CCTA datasets are sent to HeartFlow to be processed, where it takes about two to four hours to create a final 3-D image with measurements.
HeartFlow is working on software to place virtual stents in the coronary tree to estimate how revascularization of a lesion will affect coronary flow, said Campbell Rogers, M.D., FACC, chief medical officer of HeartFlow and former chief scientific officer and global head of research and development at Cordis Corp. He said the tool might be used to better plan percutaneous coronary interventional (PCI) procedures in a virtual environment.
HeartFlow’s FFR-CT software has European CE mark clearance. Rogers said the company hopes to submit for U.S. Food and Drug Administration (FDA) review in the coming months.
“Looking carefully at this fractional flow technology is something we need to be doing,” said Daniel Berman, M.D., FACC, chief of cardiac imaging and nuclear cardiology at the Cedars-Sinai Heart Institute, Los Angeles. “Evaluation of intermediate stenosis is a problematic area for CT, and this will help improve predicting what will be found in the cath lab so we can better assess patients.”
CT Perfusion Imaging
Nuclear myocardial perfusion imaging has traditionally been the gatekeeper modality to determine which patients are sent to the cath lab. However, CT may eventually challenge nuclear, offering a single scan for both detailed anatomy and function.
Several manufacturers have developed CT perfusion analysis software, which maps levels of contrast present in tissue. Low contrast attenuation in the myocardium correlates to areas of ischemia or infarct. These maps can be viewed as color-coded areas in multiplanar images or on 3-D models of the left ventricle.
TeraRecon said it plans to release a 3-D vessel overlay on its perfusion map of the left ventricle to quickly identify where ischemia originates.
CT perfusion imaging is current handicapped by the lack of validation. Clinical data gathered so far is limited to smaller, single-center studies, said Ricardo Cury, M.D., FSCCT, medical director of cardiac imaging, Baptist Hospital of Miami. He explained there are no standardized protocols for the imaging procedure, so centers examining this method all image patients differently. Artifacts in CT scans might also appear as perfusion defects, which is something he said users need to consider when evaluating these images.
Advanced Visualization of the Coronaries
Mild plaques are the cause of many heart attacks, not heavily stenosed lesions, Min said. He explained CT technology is getting to the point where it can help evaluate the composition of plaques, vessel remodeling and the level of stenosis, leading to virtual vessel biopsies.
All the major CT system manufacturers and third-party advanced visualization software vendors offer plaque assessment software. They all classify plaque make up based on the Hounsfield unit (HU) returns in each pixel of the CT image matrix. The software can auto-shade or outline the vessel wall, patent lumen (highlighted by contrast) and the different types of plaque detected. The basic detection level on all the software on the market is for calcified, non-calcified and mixed plaques.
The CT plaque characterizations do have some correlation to virtual histology intravascular ultrasound (VH-IVUS), but it is still considered crude by some users. One area of interest is the development of technology to better evaluate “napkin ring” plaques, so called because they appear as a light ring with a dark center. These are believed to represent a large lipid pool plaque, typical of vulnerable plaques. If image quality improves and CT plaque characterization is validated in clinical trials, it could offer a paradigm shift in how patients are screened and monitored for coronary artery disease noninvasively. It might also offer a noninvasive test to preemptively identify vulnerable plaques so they can be treated prior to rupturing. The same prediction has been made about VH-IVUS and intravascular optical coherence tomography (OCT), but CT would offer a less expensive and more practical noninvasive imaging method.
CT Required for TAVR
CT is expected to see increasing use for transcatheter aortic valve replacement (TAVR) patient assessment and procedural guidance.
Clinical data shows up to 12 percent of patients have moderate to severe paravalvular regurgitation following implant of the Sapien valve, said Jonathon Leipsic, M.D., FRCPC, FSCCT, chairman of the department of radiology for Providence Health Care and vice chairman of research for the department of radiology at the University of British Columbia, Canada. He explained this is due to the valve being round and the aortic annulus in many patients being elliptical or irregularly shaped. This can be fixed by oversizing the implantable valve, which can be assessed with pre-procedural imaging. For this reason, Leipsic's institution oversizes the devices and does not fully inflate the balloon, so there is room to expand the valve further it there is paravalvular leak.
Leipsic said CT is better than ultrasound to more accurately assess annulus sizing.
Reproducibility is also key with CT, instead of relying on the skill level of the echocardiographer, where there can be a wide operator variation.
CT is needed to assess the vessels from the femoral artery to the aortic root to determine if a femoral or transapical access route should be used. In addition, he said the 3-D reconstruction of the aortic arch and aortic root greatly aids proper alignment for the deployment of the Sapien in the proper landing zone.
Software is now available from Siemens and GE to overlay this CT image on live angiography. At SCCT, Philips introduced similar TAVR applications for its IntelliSpace Portal.
As the C-arm gantry moves, the CT image rotates with it. It displays a basal ring (drawn between the lower most points of aortic root cusps), which, when aligned, form a line instead of a circle. This shows there is perfect perpendicular alignment with the root, the view that is needed to proper valve alignment. Software from the angiography system vendors can also integrate CT images showing the best angle for view of the aortic valve. The view shown can be transferred automatically for the C-arm to rotate into the position to show the same view under fluoroscopy.
References:



 May 12, 2020
May 12, 2020