When I began practicing as an electrophysiologist in 1994, the dream of successfully using cardiac catheter ablation to restore a normal rhythm to patients with
atrial fibrillation (AFib) in a lab setting was just that: a dream.[1] Today, nearly 6 million Americans live with AFib, and that number is expected to nearly double by 2030.[2,3] I have been fortunate in my career to witness the introduction and aid in the design of novel technologies to meet the treatment demands created by the surge of AFib diagnoses. But there is more that must be done to improve our ability to detect and map rhythm abnormalities. At Boston Scientific, we are more motivated than ever to close that gap and improve treatment for those living with heart rhythm disorders.
Where We Have Been in Cardiac Mapping
Electroanatomical mapping has come a long way in the last 70 years. Early technology was simple, with each device performing only a single task. In the 1950s, physicians and researchers were learning how to record heart rhythms with electrocardiograms to locate arrhythmias.[4,5] In the 1970s, catheter-based imaging and 2-D ultrasound images helped better visualize heart anatomy, but pinpointing the precise location of the rhythm abnormality remained a challenge.[6,7] It was a shift to increasing usage of catheter ablation over other treatment options in the 1980s that led to the advent of combined mapping and navigation.[8,9]
In the 1990s, new 3-D systems provided more accurate data by showing heart anatomy and electrical activity together, enabling catheter localization in real time.[10-13] These advances were particularly valuable for patients with complex arrhythmias or complicated cardiac anatomy.[14] Our vision of the heart and its rhythms improved from 20/100 to 20/40 — better than before, but still not perfect.
Where We Are Heading in Cardiac Mapping to Meet the Needs of Physicians
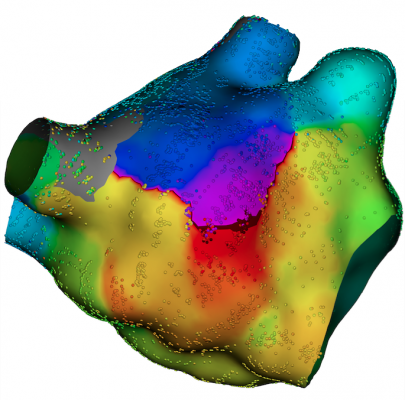
The 21st century launched us into a new era for cardiac mapping, with technological advances now emphasizing visualization in high definition and treatment with high precision. The hardware and software of new mapping systems are being designed in concert, allowing the mapping procedure to be better coordinated and automated. Unlike previous systems, the Boston Scientific Rhythmia HDx Mapping System rapidly and automatically generates 3-D maps of any chamber of the heart to help locate and treat the source of rhythm abnormality. The technology allows electrophysiologists to see the heart like never before, with 20/20 or better vision.
A recent analysis of patients with symptomatic AFib showed that compared to other mapping systems, the Rhythmia Mapping System produces higher-density voltage maps without increasing overall procedure time.[15] In fact, the technology has the ability to automatically map the heart with a speed that allows physicians greater efficiency in the EP lab and thus, more time to develop a well-informed ablation strategy.
In addition, the use of complementary technologies helps improves connectivity between the system, catheters and ablation devices, streamlining workflow in the EP lab. For example, while still an investigational device, we acquired the
Apama Medical Radiofrequency Balloon Catheter System (not yet available for sale) because its design offers the benefits of point-by-point and balloon-based ablation technologies without increasing procedure times. Our goal is for the catheter to work in connection with the mapping system for faster data collection and better procedural outcomes benefiting EP lab workflow and ultimately, patients.
We are at a pivotal point in the evolution of electroanatomical mapping, and I am in awe of how modern technology has advanced medicine. With the current momentum driving us forward, the goal is to continue to advance patient-focused solutions to a place where we can meet the needs of our patients safely, effectively, predictably and precisely.
Editor’s note: Kenneth Stein, M.D. FACC FHRS, is the senior vice president and chief medical officer for rhythm management and global health policy at Boston Scientific. Prior to joining Boston Scientific in 2009, Stein was associate director of clinical cardiac electrophysiology at Weill Cornell Medical Center and associate professor of medicine at Cornell University. He has published widely in the areas of cardiac electrophysiology with a special interest in cardiac resynchronization therapy and risk stratification for sudden cardiac arrest.
Related Content on Atrial Fibrfillation Technology Advances:
References:
1. Jais, P., Sanders, P., Hsu, L. F., Hocini, M., & Haissaguerre, M. (2005). Catheter ablation for atrial fibrillation. Heart, 91(1), 7–9. http://doi.org/10.1136/hrt.2003.030205




 April 14, 2026
April 14, 2026 









