November 18, 2011 – Medtronic and Bain Capital, a global private investment firm, today announced they have entered into a definitive agreement under which affiliates of Bain Capital will acquire Physio-Control. The stock of Physio-Control and related entities will be purchased for cash in a transaction valued at approximately $487 million.
Headquartered in Redmond, Wash., Physio-Control is a leader in professional emergency response products and services and has nearly 1,000 employees worldwide. Brian Webster, currently president, will continue to lead the company and will be named chief executive officer following completion of the transaction.
“We are pleased that Physio-Control has found another strong partner to prepare the company for the next stage of its growth,” said Omar Ishrak, chairman and chief executive officer of Medtronic. “For more than 55 years, Physio-Control has been developing technologies and designing devices that are legendary among emergency medical response professionals. With an impressive innovation pipeline and backing by Bain Capital, we believe Physio-Control will be well-positioned for growth and to continue its lifesaving mission to serve emergency responders, hospital care teams and their patients around the world.”
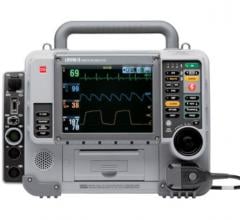
Physio-Control products, which include LifePak monitor / defibrillators, the Lucas 2 chest compression system, and the Lifenet system cloud-based data management tools, are used daily by emergency medical services (EMS) and hospital care teams worldwide. Physio-Control also provides a line of automated external defibrillators (AEDs) for lay responders’ use in workplace environments, airports, schools and communities in the case of a cardiac emergency.
“The dedicated people and innovative products of Physio-Control play a vital role in helping emergency responders and medical professionals save lives every day,” said Chris Gordon, a managing director at Bain Capital. “Physio-Control is an impressive market leader that has built an outstanding brand position and loyal customer base by providing high quality, mission-critical tools and superior service. We are extremely enthusiastic about the company’s growth prospects, and we look forward to working alongside Brian Webster and the whole Physio-Control team to support their strategic plans.”
The transaction is expected to close in the first quarter of calendar 2012, and is subject to certain regulatory and customary closing conditions.
Non-equity financing for the transaction is being provided by Citigroup Global Markets Inc. and RBC Capital Markets. Goldman Sachs is acting as financial advisor, and Skadden, Arps, Slate, Meagher & Flom LLP as legal advisor to Medtronic. Citigroup Global Markets Inc. and RBC Capital Markets are acting as financial advisors and Kirkland & Ellis as legal advisor to Bain Capital.
Medtronic expects to use a portion of the proceeds to offset any potential dilutive impact from this transaction to fiscal year 2012 non-GAAP earnings per share.
For more information: www.medtronic.com, www.baincapital.com


 March 10, 2026
March 10, 2026