May 29, 2008 – Providing objective metrics and educational feedback to medical personnel who performed CPR on a cardiac arrest patient significantly improved ventilation rate, compression rate and depth of compression, and was associated with higher initial survival rates for cardiac arrest patients, according to results published in the Archives of Internal Medicine from the Resuscitation with Actual Performance Integrated Debriefing (RAPID) trials.
“Similar to post-game analysis for professional athletes, medical responders were able to learn from their past resuscitation performance and apply this knowledge to future resuscitations,” said Dana Edelson, M.D., director of Clinical Research for the Emergency Resuscitation Center at the University of Chicago Medical Center, and co-investigator of the study. “The results of this study suggest that CPR performance review has broad applicability for improving resuscitation training.”
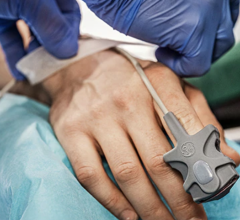
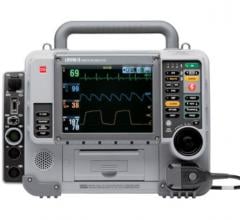
Results drawn from medical professionals participating in RAPID trials were published in the study, “Improving In-Hospital Cardiac Arrest Process and Outcomes using Performance Debriefing.” The Philips HeartStart MRx with Q-CPR Measurement and Feedback was used to record resuscitation performance during actual cardiac arrest events. Medical professionals then participated in weekly debriefing sessions to review transcripts from the prior week’s resuscitations and analyze CPR performance including chest compression rate and depth, ventilation rate, and undesired pauses in compressions.
Only in recent years has it been possible to measure CPR performance in a clinical setting. With its data collection and review capabilities, Q-CPR allows resuscitation leaders in hospitals to use valuable feedback from actual cardiac arrest cases to supplement classroom-based training methodologies for professional responders.
The trial results demonstrate that CPR performance quality improved for study participants who received post-resuscitation feedback compared to a control group of professionals who did not receive educational intervention. Specifically, the results show that:
During the educational intervention period, there was significant improvement on CPR performance measures, compared to the control period including:
o Faster chest compressions
o Deeper chest compressions
o Decreased pauses in chest compressions, in general, as well as around the time of defibrillation
o Decreased ventilation rates
These improvements in CPR performance correlated with an increased rate of initial survival (or “return of spontaneous circulation”, also termed “ROSC”) in the RAPID group.
“As the trial demonstrates, balancing the correct number of breaths with the exact depth and rate of chest compressions in real-life CPR performance is actually a very complex process with a high likelihood of human error,” said Benjamin S. Abella, M.D., MPhil, assistant professor, Department of Emergency Medicine, University of Pennsylvania, and co-investigator of the study. “A focused educational debriefing program using data provided by the HeartStart MRx with Q-CPR technology helps emergency responders deliver quality CPR consistently, even in the most stressful and fast-paced environments.”
Quality CPR and early defibrillation are inextricably linked – the combination is essential to increase the survival rate for victims of cardiac arrest. Q-CPR, available on the Philips HeartStart MRx Monitor/Defibrillator, is integrated into a monitor/defibrillator that provides real-time CPR monitoring and feedback for both chest compressions and ventilations (breaths). The technology, developed by Philips and Laerdal, is said to offer medical professionals corrective feedback on the rate and depth of chest compressions, as well as the frequency and quality of ventilations, encouraging them to adjust their technique as needed to improve CPR.
“Studies such as the RAPID trial reinforce the need to better integrate CPR solutions with early defibrillation. We believe this to be the most promising path to improving survival rates for the thousands of victims of cardiac arrest each year,” said Michael Miller, senior vice president of Cardiac Care for Philips Healthcare. “As a worldwide leader in resuscitation, Philips is dedicated to offering integrated solutions such as the HeartStart MRx with Q-CPR, to raise the bar in quality of resuscitation and ultimately improve patient outcomes.”
For more information: www.medical.philips.com


 March 10, 2026
March 10, 2026