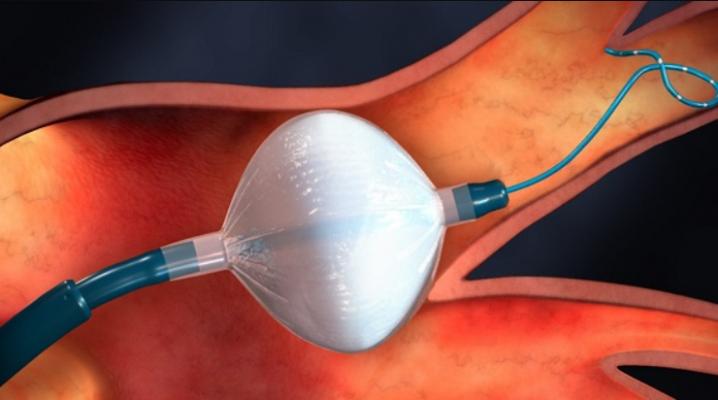
The Medtronic Arctic Front cryoballoon balloon was used a a front-line treatment of AF in the EARLY-AF trial. It was found to be better than anti-arrhythmic drugs in preventing the recurrence of abnormal heart rhythm (atrial tachyarrhythmia, atrial fibrillation), and improve patient well-being.
November 16, 2020 – Doctors from the Montreal Heart Institute (MHI) presented new data that shows catheter ablation as first-line treatment may be better than anti-arrhythmic drugs in preventing the recurrence of abnormal heart rhythm (atrial tachyarrhythmia, atrial fibrillation), and improve patient well-being.
The data were presented at the 2020 American Heart Association (AHA) Scientific Sessions and simultaneously published in the New England Journal of Medicine.[1]
Atrial fibrillation (AF) is the most common cardiac arrhythmia, affecting approximately 1 to 2% of the overall population. Treatment guidelines recommend a trial of drug therapy to maintain a normal atrial heart rhythm before catheter ablation is considered. However, these medications have only limited ability to maintain a normal heart rhythm and have substantial side effects. Catheter ablation is considered superior to anti-arrhythmic drugs in maintaining heart (sinus) rhythm and improving quality of life in patients in whom drugs have already failed but it was previously unknown whether earlier ablation could improve patient outcomes.
Data from The Early Aggressive Invasive Intervention for Atrial Fibrillation (EARLY-AF) trial found that in treatment-naïve patients with symptomatic AF, initial catheter cryoballoon ablation resulted in a significantly lower recurrence of arrhythmia compared to those who received antiarrhythmic drug therapy.
“Atrial fibrillation is associated with significant morbidity, and an estimated two-fold increase in premature death.Unfortunately, patient outcomes are worse when ablation is delayed.,” said Jason Andrade, M.D., FHRS, FRCPC, and electrophysiologist affiliated with the MHI, assistant professor of medicine, Université de Montréal and lead author of the EARLY-AF study.
“These data add to evidence that an early invasive therapy may be a more effective method to improve the lives of our patients with atrial fibrillation. In addition, this study supports the recommendation of pursuing catheter ablation an initial treatment strategy in appropriate patients,” added Dr. Laurent Macle, chief of the electrophysiology service at MHI, and co-chair of the recently published 2020 Canadian Cardiovascular Society Atrial Fibrillation guidelines.
The EARLY-AF study was a multicenter, parallel-group, randomized trial that enrolled participants with treatment-naïve symptomatic AF. A total of 303 patients were randomly assigned either to undergo cryoablation (154 patients) or to receive antiarrhythmic drug therapy (149 patients). At 1 year, a recurrence of symptomatic episodes of atrial tachyarrhythmia had occurred in 17 of 154 patients (11.0%) assigned to undergo ablation and in 39 of 149 patients (26.2%) assigned to receive antiarrhythmic drugs (hazard ratio, 0.39; 95% confidence interval [CI], 0.22 to 0.68; P<0.001). Patients treated with initial ablation had a significantly greater improvement in multiple measures of quality of life, were more likely to be asymptomatic, and had a lower rate of hospitalization. Serious adverse events occurred in 5 patients (3.2%) who underwent ablation and in 6 patients (4.0%) who received antiarrhythmic drugs.
The study was conducted at 18 centers in Canada and the full data can be found at: https://www.nejm.org/doi/full/10.1056/NEJMoa2029980
Find more AHA late-breaking studies, news and video
Reference:


 November 14, 2025
November 14, 2025