March 25, 2026 — Royal Philips has received U.S. Food and Drug Administration (FDA) 510(k) clearance for EchoNavigator R5.0 with DeviceGuide, an AI-powered software solution that assists physicians during one of interventional cardiology’s most technically demanding procedures – repairing leaking mitral valves through a minimally invasive approach.
DeviceGuide was developed in close collaboration with Edwards Lifesciences, aligning Philips’ imaging and AI expertise with Edwards’ expertise in valvular heart therapy development. Together, the companies have innovated image guidance of the mitral transcatheter edge-to-edge repair (M-TEER) workflow to make these complex minimally invasive heart valve repair procedures more intuitive and streamlined.
DeviceGuide will be showcased at the American College of Cardiology (ACC) 2026 meeting in New Orleans.
Transforming Treatment
A leaking mitral valve, known as mitral regurgitation (MR), affects more than 35 million adults worldwide and over 2 million in the U.S.3,4 Minimally invasive transcatheter techniques such as M-TEER offer an alternative to open heart surgery in selective patient populations.
During M-TEER procedures, physicians make a small skin incision to access a vein which allows the introduction of a catheter to the heart to deliver a repair device to the diseased mitral valve. Because the mitral valve is inherently a complex and heterogeneous structure, transcatheter repair of the valve requires experienced physicians positioning the device delivery system, and physicians guiding the placement of the device. In guiding and positioning the repair device, the intraprocedural heart team must interpret both X-ray and ultrasound images on multiple screens, communicate and coordinate movements between two operators, and make precise adjustments to grasp the moving valve leaflets and then confirm the result in real time. The process demands accuracy, coordination and experience from the entire team — and this is where DeviceGuide can help with navigation guidance.
AI Assistance
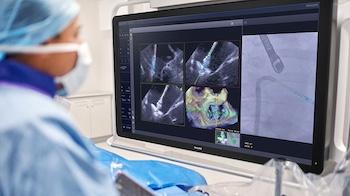
Built on Philips’ EchoNavigator echo-fluoro fusion technology, which combines live echocardiography images from Philips EPIQ CVxi cardiovascular platform with live X-ray images from Philips Azurion image-guided therapy system, DeviceGuide brings real-time AI guidance directly into the procedure room.
The software’s AI algorithm automatically tracks and visualizes the Edwards PASCAL Ace mitral valve repair device, combining live ultrasound and X-ray images into a single, integrated view. This helps clinicians navigate and position the device with greater clarity and confidence.
“The AI software serves as an assistive tool; the physician always remains in control. This isn’t about replacing expertise – it’s about amplifying it,” explains Dr. Atul Gupta, Chief Medical Officer Diagnosis & Treatment at Philips. “By embedding AI into the procedure, DeviceGuide gives physicians an extra pair of eyes, helping them treat more patients safely and confidently.”
Collaboration with Clinical Partners
In developing DeviceGuide, Philips and Edwards worked closely with investigational sites in Europe and the U.S., including a team at the Structural Heart and Valve Center at NewYork-Presbyterian/Columbia University Irving Medical Center, led by interventional cardiologist Susheel Kumar Kodali, MD, director, and Rebecca T. Hahn, MD, director of interventional echocardiography.
“In helping to guide mitral repair procedures, one of my roles as an echocardiographer is to help the interventional cardiologist understand the complex anatomy of the valve which will determine the orientation, trajectory and position of the repair device relative to the target and the surrounding structures,” said Dr. Hahn. “Since AI auto-aligns imaging to the device in real time and continuously informs the interventionalist about the location of the device in space on the imaging screen, it minimizes unnecessary repositioning of the imaging window, streamlines procedural guidance and may improve the precision of device implantation.”
“DeviceGuide provides me with a visual overlay, trajectory line and orientation line of the therapy device in both live 3D echo and fluoroscopic images during mitral valve repair procedures,” said Dr. Kodali. “Having a single, intuitive presentation of real-time target, orientation and auto device-aligned views simplifies this procedure and improves team communication.”
Real-Time Support
“Structural heart procedures are among the fastest-growing areas in cardiology, and also among the most complex,” said Mark Stoffels, Business Leader, Image Guided Therapy Systems at Philips. “By assisting physicians with real-time visualization and navigation inside the beating heart, DeviceGuide helps them manage that complexity, and perform procedures more confidently, with the ultimate goal to treat more patients effectively. It fits seamlessly into cath-lab workflows and gives the entire team a shared view of the procedure, improving coordination and confidence.”
The Philips’ Cardiology Ecosystem
DeviceGuide extends Philips’ connected cardiology portfolio that helps physicians care for heart patients from diagnosis through recovery. It also represents a step toward Philips’ vision of the AI-powered cath lab of the future where imaging, devices, and real-time data are intelligently connected to reduce procedural complexity and improve consistency. In hospitals, Philips systems already bring together ultrasound, X-ray, and real-time data into a unified workflow to guide minimally invasive procedures. Building on this foundation, intelligent software solutions such as DeviceGuide help care teams navigate complex structural heart interventions with greater clarity and coordination, serving as an assistive “copilot” in the procedure room.
After treatment, Philips connected monitoring tools extend care beyond the cath lab, helping care teams detect complications early and support recovery. Together, these innovations create a connected cardiology ecosystem that helps care teams diagnose earlier, treat less invasively, improve procedural efficiency, and support recovery – improving outcomes and experiences for heart patients everywhere.
Availability
DeviceGuide enabled by EchoNavigator is FDA 510(k) cleared in the United States. Commercial availability is subject to market release and applicable regulatory requirements. DeviceGuide is currently intended for use with the Edwards PASCAL Ace Mitral Valve Repair System. Availability outside the U.S. varies by country and regulatory status.
A recent first-in-human publication in JACC: Case Reports describes early clinical experience using DeviceGuide for AI-supported imaging guidance during mitral transcatheter edge-to-edge repair procedures.
Read more on how Philips DeviceGuide works here.
Sources
-
DeviceGuide is currently intended for use only with the Mitral TEER Therapy Device (Edwards PASCAL Ace); Hahn RT, Biaggi P, Corti R et al. Mitral Transcatheter Edge-to-Edge Repair Using Novel Augmented Imaging Software. JACC: Case Reports 2025;30:106160.
-
DeviceGuide assists physicians in visualizing and navigating the repair device within the heart. It supports, but does not perform, the therapeutic procedure itself; Biaggi P, Corti R, Gaemperli O et al. Artificial intelligence based fusion imaging streamlining mitral transcatheter edge-to-edge repair. European Heart Journal - Imaging Methods and Practice 2026;4.


 October 21, 2025
October 21, 2025 




