December 19, 2016 — BioTrace Medical Inc. announced the first commercial use of the company’s Tempo Temporary Pacing Lead since U.S. Food and Drug Administration (FDA) 510(k) clearance in October.
The first cases involved patients undergoing transcatheter aortic valve replacement (TAVR) procedures and were performed by James Harkness, M.D., interventional cardiologist, and Brian K. Whisenant, M.D., medical director of the Structural Heart Disease Program at Intermountain Medical Center in Salt Lake City, Utah, and Susheel Kodali, M.D., director of the Heart Valve Program at Columbia University Medical Center/New York Presbyterian Hospital.
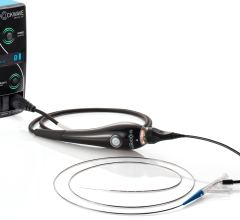
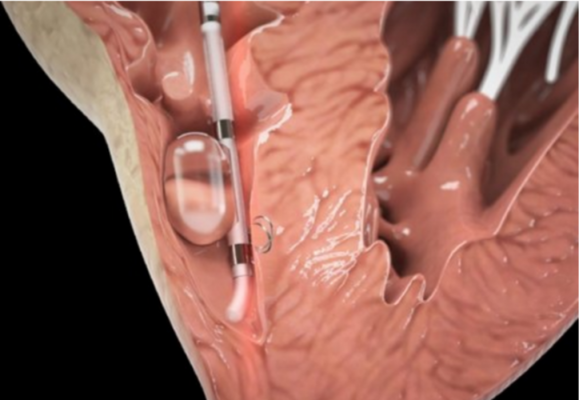
BioTrace Medical’s Tempo Lead is for use in procedures in which temporary pacing is indicated, including TAVR and electrophysiology (EP) procedures. The lead is designed for secure and stable cardiac pacing with the goal of reducing complications and allowing patients to ambulate sooner after procedures.
“I am pleased to now have an innovative new option for temporary pacing for my patients,” said Whisenant. “The lead reliably allows stable and secure placement, and is easy to use. The Tempo Lead is the new standard of care for patients who need temporary pacemaker support.”
“BioTrace’s temporary pacing leads are an exciting advance over existing technologies, which can cause complications that result in poor clinical outcomes and longer hospital stays,” said Martin B. Leon, M.D., director of the Center for Interventional Vascular Therapy at Columbia University Medical Center/New York Presbyterian Hospital, and a member of BioTrace Medical’s Scientific Advisory Board. “The Tempo Lead is designed to alleviate the risks associated with lead dislodgement and inconsistent pacing, providing a safer option for patients.”
Temporary leads are used in more than 350,000 procedures each year, a number that is growing rapidly as the population ages and TAVR becomes increasingly common. The temporary pacing lead, a small catheter with two electrodes, is placed in the right ventricle of the heart through a vein in the groin or neck. The lead is then connected to an external pacemaker allowing a physician to monitor and control a patient’s heart rate for several days.
The Tempo Lead features a novel active fixation mechanism, bipolar electrodes and a soft tip.
Read the article “First U.S. TAVR Patients Treated With Temporary Pacing Lead.”
Safety and efficacy results of the first-in-human study of the Tempo Lead were presented in October at the annual Transcatheter Cardiovascular Therapeutics (TCT) conference in Washington, D.C. The multi-center study clearly demonstrated the safety of the Tempo Lead, with no device-related adverse events, dislodgements, sustained ventricular arrhythmia, or cardiac perforations. The device was successfully positioned in 23 patients (92 percent), with two patients having unsuitable anatomy. Pacing with the Tempo Lead was successful in all treated patients with no loss of pace capture or lead dislodgement of the device from within the heart.
The design of conventional temporary pacing leads can cause serious complications. Current temporary leads can be easily dislodged from the heart, which may result in loss of pacing, with serious consequences for the patient. As a result, patients are often limited to bed rest for the duration of temporary pacing lead placement, delaying ambulation and thereby increasing length of stay in costly hospital units such as intensive care. In addition, current leads risk perforation of the heart wall, which may lead to pericardial effusion and tamponade, in which blood from within the heart escapes into the sac surrounding the heart, leading to potentially life-threatening cardiac compression.
BioTrace Medical, Inc., a venture backed company based in San Carlos, Calif., is dedicated to reinventing temporary pacing to improve patient outcomes and reduce hospital costs.
For more information: www.biotracemedical.com


 December 20, 2023
December 20, 2023