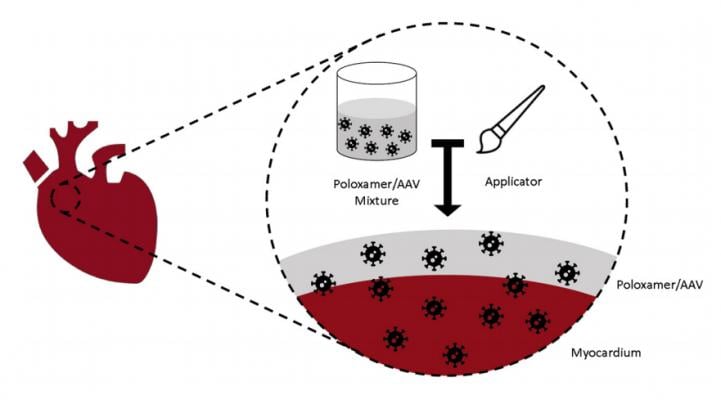
April 19, 2021 — National Heart, Lung, and Blood Institute (NHLBI) has awarded a grant for $462,689 to Rhythm Therapeutics Inc. (RTI) to complete the phase 1 segment of development of a novel gene therapy to treat atrial fibrillation (AF). It involves using electroporation to create temporary openings in cardiac cells to deliver therapeutic transgenes targeting the underlying molecular mechanisms of AFib.
Upon successful completion of the phase 1 segment (projected year-end 2021), RTI will be eligible to receive an additional $3,212,600 in NIH funding to carry out the Phase 2 segment, which will include determining optimal dose of gene therapy required to attenuate established electrical remodeling in AF, determining the duration of gene efficacy, and assessing the toxicology profile of targeted genes.
“AF is the most common heart rhythm disorder, affecting 6 million people in the U.S. and 33 million worldwide,” said Rishi Arora, M.D., FHRS, FAHA, CEO of Rhythm Therapeutics and professor of medicine, clinical cardiac electrophysiology at Northwestern University, Feinberg School of Medicine. “Existing standard of care treatments including drugs and ablation have suboptimal efficacy for patients with AF. This is especially the case for patients with more advanced AF or ‘persistent’ AF, which constitutes nearly half of the AF patient population. Improving patient care and avoiding readmissions for high-burden patients is increasingly becoming a focus for payers and healthcare systems.”
Under Arora’s leadership, Rhythm Therapeutics has identified major molecular mechanisms contributing to AF, identified trans-genes to selectively target these mechanisms, and developed a novel method of facilitating gene delivery via electroporation. This has decreased AF in clinically relevant, large animal models of atrial fibrillation.
“The use of targeted electroporation to deliver gene therapy to the heart is an exciting addition to the field of gene therapy,” stated grant principal investigator and gene therapy industry veteran Robert C. Moen, M.D., Ph.D. “We believe this grant award enables RTI to complete important steps necessary before its promising approach can be tested in the clinic.”
“With support from this valued award, Rhythm Therapeutics has the opportunity to revolutionize the treatment of persistent atrial fibrillation via our proprietary gene therapy platform with the goal of reducing mortality, and improving the quality of life for those suffering with AF worldwide,” said Arora.
Following projected receipt of the SBIR award in May 2020, RTI is planning to move company operations to Portal Innovations, a life sciences focused venture development engine located in Chicago's Fulton Market neighborhood providing crafted capital in the form of wet lab & office space, seed investments, managerial expertise and an extensive partner network to promising early stage life science companies developing life saving technologies for patients.
Rhythm Therapeutics Inc., a privately held biotechnology company based in Chicago, is dedicated to delivering the promise of gene therapy for treating serious cardiovascular diseases such as atrial fibrillation.
Atrial fibrillation is associated with a 4-5 times higher risk of stroke and heart failure and is a significant health issue worldwide.
For more information: www.rhythmtherapeutics.com


 April 14, 2026
April 14, 2026 









