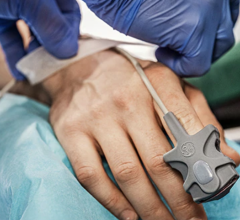
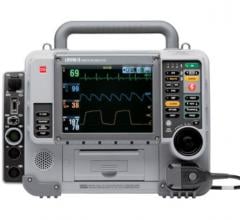
July 23, 2013 — Zoll Medical Corp has announced that survival from one of the leading causes of death in the United States, out-of-hospital cardiac arrest, more than doubled from 26 percent to 56 percent when paramedics in Mesa, Ariz., utilized Zoll’s cardiopulmonary resuscitation (CPR) feedback technology in combination with scenario-based training. The findings also showed that high-quality CPR was associated with significantly improved neurologically intact survival.
Fifty-six percent of witnessed cardiac arrest victims in Mesa survived with good functional outcomes in this study, which used Zoll’s defibrillators with Real CPR Help. The GPS-like technology allows rescuers to “see” and receive real-time audiovisual feedback on the quality of their CPR, including the depth and rate of their chest compressions.
“We believe that CPR is the most important factor in cardiac resuscitation. But it’s not just about doing CPR, it’s about doing CPR right,” said study author Ben Bobrow, M.D., who is the medical director for the Arizona Department of Health Services’ Bureau of EMS and Trauma System and professor of emergency medicine at Maricopa Medical Center, University of Arizona College of Medicine Phoenix.
“The fact that more than twice as many people returned home to their families shows the great importance of CPR quality and the huge potential CPR feedback technology has in improving outcomes from sudden cardiac arrest,” Bobrow added. “These results clearly demonstrate how critical it is to measure CPR quality during each and every cardiac resuscitation.”
ZOLL’s Real CPR Help uses audio and visual prompts to guide the rescuers to the actual depth of 2 inches and rate of 100 chest compressions per minute as recommended in the 2010 American Heart Association (AHA) Guidelines. The Mesa study validates the consensus statement published by AHA, of which Bobrow is a co-author, which recommends the use of some means to measure CPR quality during all resuscitations both inside and outside the hospital.
For more information, visit www.zoll.com


 March 10, 2026
March 10, 2026