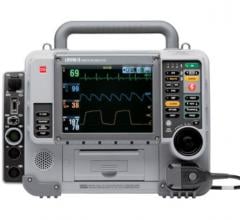
August 6, 2008 - ZOLL Medical Corp. said today the ZOLL M Series defibrillator has been installed in a number of high profile EMS centers and hospitals in the People’s Republic of China.
More than 250 M Series have been installed in both ambulances and hospitals in Beijing over the past 18 months to service the Summer Olympic Games and help improve the city’s emergency service levels. In Shanghai, the largest city in China, the M Series has been selected to equip the city’s newly-expanded ambulance service. To date, the Shanghai Medical Emergency Service Center has placed 145 M Series into service in the metropolitan area.
In addition, ZOLL was awarded a Beijing government public tender to install 191 AED Plus units into Community Medical Service Centers in Beijing City. This project marks the first time that AEDs are being made available to local residents in China through health centers and clinics. This is an integral part of China’s new healthcare system, which is designed to provide healthcare and first rescue response at the community level.
Installation of the M Series and the AED Plus follows an earlier announcement that several major hospitals and EMS services in China have installed ZOLL’s AutoPulse Non-invasive Cardiac Support Pump. Four AutoPulse units will be onsite with Shenyang EMS during the Olympic soccer games taking place in that city next month. Earlier in the year, ZOLL said 80 AED Plus units were being installed in newly-built Terminal 3 at Beijing International Airport, the gateway terminal for the 2008 Beijing Summer Olympics.
The company said the small, rugged M Series is an easy-to-use portable unit. It has straightforward controls to provide everything for cardiac resuscitation: defibrillation, external pacing and advanced monitoring capabilities. Its 12-lead ECG data transmission provides clinicians with information from the field to assess the patient’s condition before arriving at the hospital for improved diagnosis and treatment of patients.
For more information: www.zoll.com.


 March 10, 2026
March 10, 2026