
Jan. 16, 2026 — MemorialCare Heart & Vascular Institute has launched the FDA-approved Symplicity Renal Denervation (RDN) procedure, a minimally invasive treatment now available at Long Beach Medical Center, Orange Coast Medical Center and Saddleback Medical Center. According to the American Heart Association, an estimated 17–20% of Americans with hypertension — about 10 million people — have resistant hypertension, where blood pressure remains high despite multiple medications. The procedure can reduce major cardiovascular events by 20%, stroke by 27% and heart failure by 28% in patients with resistant hypertension.
"This procedure offers a new pathway for patients who haven't found success with traditional treatments," says David Shavelle, M.D., FACC, FSCAI, chief of cardiology, MemorialCare. "It's a safe, effective option that works in harmony with existing care plans to help patients achieve better blood pressure control. We're proud to bring this innovative therapy to our communities and help patients regain control and peace of mind."
High blood pressure affects nearly half of U.S. adults and is a leading cause of heart disease, stroke, and kidney failure. While many individuals are able to manage their blood pressure with medications and lifestyle adjustments, an estimated 17–20% of patients experience resistant hypertension, a condition in which blood pressure remains high despite taking three or more antihypertensive medications.
Resistant hypertension is more common among certain populations, including older adults, Black patients and individuals with chronic conditions such as diabetes, chronic kidney disease, and obesity. Black adults are nearly 50% more likely to develop resistant hypertension compared to white adults. These groups face a significantly higher risk of stroke, heart failure, and major cardiovascular events, making innovative treatments like renal denervation especially critical.
The procedure works by delivering targeted radiofrequency energy to overactive nerves surrounding the renal arteries — nerves that contribute to elevated blood pressure — helping the body naturally regulate blood pressure. Clinical trial data from Medtronic shows that a 10-mmHg reduction in office systolic blood pressure can reduce major cardiovascular events by 20%, stroke by 27% and heart failure by 28% in patients with resistant hypertension. According to the American Heart Association, resistant hypertension increases cardiovascular mortality by up to 56% compared to controlled hypertension, making this advancement a critical option for patients whose blood pressure remains uncontrolled despite medication and lifestyle changes.
"Clinical studies have shown that renal denervation can significantly lower both systolic and diastolic blood pressure, support long-term blood pressure stability, and reduce the need for multiple medications," says Dr. Shavelle. "This minimally invasive procedure complements existing treatment plans and offers a safe, effective option for patients who cannot tolerate certain blood pressure medications."
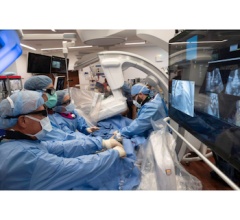
The Symplicity Spyral RDN system, developed by Medtronic, is a minimally invasive therapy that uses a thin, flexible catheter to deliver targeted radiofrequency energy to overactive nerves surrounding the renal arteries — nerves that disrupt communication between the kidneys and the brain and contribute to elevated blood pressure. Through a small incision near the upper leg or groin, the catheter is guided to the renal arteries, where energy is applied to specific spots along the artery wall. This calms the nerves without damaging the artery, allowing the body to naturally regulate blood pressure. The treatment typically takes about an hour, and most patients return home the same day or after a short observation period, without the need for surgery or permanent implants.
MemorialCare's launch of the renal denervation blood pressure procedure reflects its commitment to offering advanced cardiovascular care. The system-wide implementation is part of a broader strategy to expand access to innovative therapies and improve outcomes for patients with complex cardiovascular conditions.
To learn more visit memorialcare.org/Heart

 February 11, 2026
February 11, 2026 

