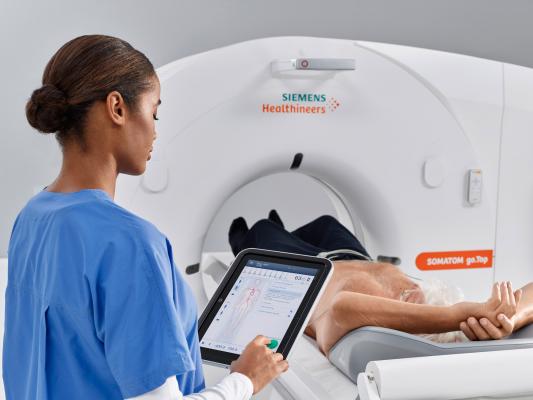
March 14, 2019 — Siemens Healthineers will introduce the Somatom go.Top Cardiovascular Edition, a new version of its established computed tomography (CT) system, at the 68th Annual Scientific Session and Expo of the American College of Cardiology (ACC), March 16-18 in New Orleans. The Somatom Go.Top Cardiovascular Edition is designed to deliver personalized patient dose control in all types of routine cardiovascular imaging. The cost-effective, 128-slice scanner offers outpatient cardiology offices as well as hospitals access to not just coronary CT angiography (CCTA), but also advanced tests, including the HeartFlow FFRCT (fractional flow reserve computed tomography) Analysis.
The system’s tablet-based mobile workflow not only simplifies and streamlines cardiac image acquisition, but it also enables the technologist to remain with the patient to help ensure comfort and calm – key factors for a successful cardiac exam. The CARE kV feature automatically selects the optimal kV setting for each patient with the precision of 10 kV increments, for personalized reduced radiation dose control as well as improved contrast enhancement. The system also has a fast native temporal resolution required for optimal visualization of coronary arteries.
The HeartFlow FFRCT Analysis, a personalized cardiac test developed by HeartFlow Inc., enables clinicians to better evaluate the impact a blockage has on blood flow and determine the best treatment for patients.
GO technologies help reduce workflow inconsistencies and variabilities that can result in poor image quality. For example, the Check&GO algorithm helps identify problems associated with anatomical coverage and contrast media distribution as they occur.
For more information: www.usa.healthcare.siemens.com


 April 02, 2026
April 02, 2026 









