Jordan Merecka (right), received a new heart Oct. 29, after 160 days with a Syncardia Total Artificial Heart.
March 5, 2012 — SynCardia Systems Inc., manufacturer of the world’s first and only U.S. Food and Drug Administration (FDA), Health Canada and Conformité Européenne (CE) approved Total Artificial Heart, announced that 18-year-old Jordan Merecka was successfully bridged to a heart transplant at Texas Children’s Hospital in Houston, Texas, after 160 days of support with the SynCardia temporary Total Artificial Heart.
“Before I got the Total Artificial Heart, I had a lot of chest pain. I could only walk about 100 yards before I was out of breath and had to stop because I couldn’t go any farther,” said Jordan. “With the Total Artificial Heart, I could walk again, go outside, and I had no more chest pain. I almost felt back to my old self. Now that I have my new donor heart, it's just getting back to a normal life step by step.”
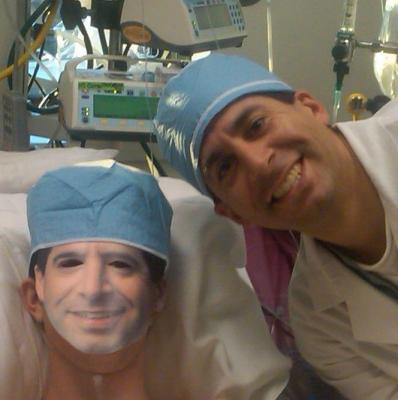
Just two days after his Oct. 29 transplant, for Halloween, Jordan masqueraded as his surgeon, David L.S. Morales, M.D. "Jordan was a little more than 24 hours out of a 12-hour surgery and he's the one cracking jokes."
Jordan made headlines in August 2011 when he became the first teen in the U.S. to be discharged from the hospital using the Freedom portable driver to power his Total Artificial Heart. Weighing 13.5 pounds, the Freedom driver is the world’s first wearable power supply for the SynCardia Total Artificial Heart. The Freedom driver is CE approved for use in Europe and undergoing an FDA-approved Investigational Device Exemption (IDE) clinical study in the U.S.
“It was a really good feeling getting to go home again,” said Jordan. “I’d go on little walks and go fishing with my friends. My sisters and my brother were really happy that I was back and that Mom and Dad were home so they weren’t alone all the time.”
Jordan was born with multiple congenital heart defects, including a “reversed” heart (dextrocardia) and his heart vessels backwards (corrected transposition of the great arteries). While awaiting a transplant, he was admitted to the hospital in April 2011 suffering from end-stage biventricular heart failure. The following month, his vital organs began to fail and he was placed on a ventilator. To save Jordan's life, on May 22, 2011, his doctors performed Texas Children’s first implant of the Total Artificial Heart.
For more information: www.syncardia.com


 June 19, 2024
June 19, 2024 









