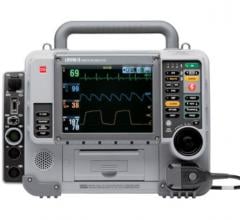
ZOLL Medical Corp. has received FDA clearance to market and sell the ZOLL M Series with Real CPR Help technology. The device enables rescuers to instantly see and hear how well they are performing the rate and depth of CPR chest compressions.
The M Series is the Company’s best-selling product to date. The approval extends Real CPR Help to ZOLL’s complete defibrillator product line.


 March 10, 2026
March 10, 2026