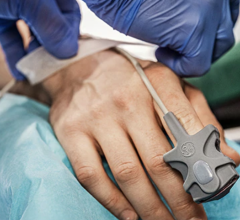
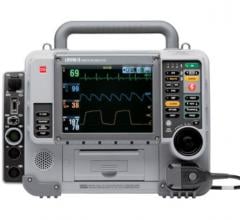
March 27, 2009 - The Philips HeartStart MRx monitor/defibrillator now includes new clinical decision support tools, updated data management solutions and next-generation Q-CPR technology to help reduce critical time to lifesaving treatment. The recommended time to treatment for patients suspected to be suffering from a class of heart attack called ST-segment elevation myocardial infarction (STEMI) — typically with balloon angioplasty performed in the hospital’s catheterization lab — is less than 90 minutes from “door to balloon.” The company says its new enhancements support this objective by helping caregivers triage patients in the pre-hospital setting, which can reduce critical time to lifesaving treatment. The HeartStart MRx monitor/defibrillator offers Philips’ new DXL ECG Algorithm, which enables analysis of 12-lead electrocardiogram (ECG) data. Unlike other 12-lead algorithms, the DXL Algorithm identifies the STEMI-CA (Culprit Artery) and provides critical values. Philips STEMI-CA pinpoints the probable congested artery, while critical values highlight any of four distinct life-threatening conditions requiring immediate clinical attention. In addition, enhanced gender-specific criteria are said to provide valuable information for diagnosing cardiac emergencies in women. These tools provide user-friendly data designed to speed triage, quickly mobilize resources and more efficiently prepare for treatment in the cath lab. “In suspected STEMI situations – where minutes literally can save lives – the diagnostic-specificity provided by the new DXL Algorithm on the HeartStart MRx enables us to better prepare for a patient’s arrival and treatment,” said Dr. Raymond Fowler, professor of Emergency Medicine at the University of Texas Southwestern Medical Center and chief of Operations for the Dallas Metropolitan (Bio Tel) EMS System. “The new tools available on the MRx provide additional insight into the condition of the patient while en route to the hospital and help us make faster and better informed treatment decisions.” Two additional tools available on the HeartStart MRx are the Philips Acute Cardiac Ischemia Time Insensitive Predictive Instrument (ACI-TIPI) and Thrombolytic Predictive Instrument (TPI) algorithms, which enable medics to alert and prepare the hospital ahead of the arrival of a suspected STEMI patient and/or bypass the closest hospital for a cardiac specialty center. ACI-TIPI provides a percentage score indicating the probability that the patient is experiencing acute cardiac ischemia. TPI provides mortality predictions in considering the use of thrombolytics to treat STEMI patients when percutaneous coronary intervention (PCI) is not readily available. The MRx also now features the next-generation Q-CPR Measurement and Feedback tool as well as support system- or institution-wide quality improvements using robust retrospective data review capabilities. This update features the innovative new Q-CPR meter, which displays visual feedback directly where the caregiver is focused —o n the patient’s chest. Q-CPR remains the only tool currently available that allows professionals to get feedback on ventilations as well as compressions. The first generation Q-CPR is supported by more published data than any other CPR quality-improvement tool, including studies in the Archives of Internal Medicine and Journal of the American Medical Association. The enhanced HeartStart MRx also features multiple data management solutions designed to streamline the flow of patient information. Specifically, Philips’ Periodic Clinical Data Transmission collects ECG and vital patient information in the field and periodically sends this data to the receiving hospital, enabling physicians to analyze and track patients ahead of their arrival. Only the MRx has a built-in Ethernet port that allows for IP-based transfer of code event summaries at LAN speed. New file-forwarding software called Data Messenger has also been added to automate the transfer of clinical care event summaries for reliable review by medical directors. "As an EMS medical director, it’s important to me that my team learn from each cardiac event and apply this knowledge to future cases in order to improve patient care,” Dr. Fowler said. "With its new virtually real-time data transfer capabilities, the HeartStart MRx truly raises the bar for the market. Within one minute, I’m able to download clinical transport data from each case for both quality review and training purposes, allowing the MRx to be returned to the field where it’s needed most.” For more information: www.philips.com


 March 10, 2026
March 10, 2026