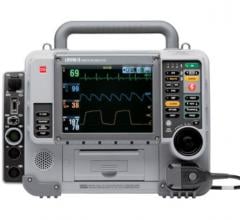
March 12, 2012 — Philips introduced the tactically enhanced HeartStart MRx monitor-defibrillator for emergency medical services (EMS). Designed in collaboration with EMS professionals, the latest enhancements will further help professional responders improve and save lives.
EMS professionals require easy to use, technically advanced monitors to focus on patient care. With a true commitment to meaningful innovation, Philips is introducing numerous improvements in the HeartStart MRx. The newest innovations are based on extensive feedback from EMS professionals. Each enhancement is designed to help professional responders optimize workflow and include:
· Tactical gray color option designed to show less dirt and wear over time
· Automatic lead switching, which allows users to focus on the patient, not the monitor
· Improved electrocardiogram (ECG) performance, including new “ruggedized” EMS lead sets
· Flexible event summary print options for enhanced workflow
· Enhanced event markers, with the ability to enter drug dose and unit of measure
· Auto switching from demand-mode to fixed-mode pacing if primary ECG lead is lost
The HeartStart MRx combines industry-leading monitoring technology with diagnostic measurements and Philips’ patented defibrillation therapies in a tough, lightweight device. The HeartStart MRx provides several advanced features important to EMS professionals, including Philips’ DXL 12-Lead ECG algorithm, a large color display, fast time to shock and the longest battery operating time for ECG monitoring among eight leading monitor/defibrillators, according to published manufacturer’s specifications. Additionally, Q-CPR, a cardiopulmonary resuscitation (CPR) measurement and feedback tool, is available as a fully integrated option.
For more information: www.philips.com/mrx-ems


 March 10, 2026
March 10, 2026