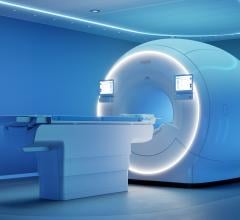
The FerrAlert Ferromagnetic Incident Log Manager (FILM) device is designed to work exclusively with Kopp Development's FerrAlert Halo II Plus MRI entryway system, pictured above. Image courtesy of Kopp Development Inc.
November 17, 2015 — Kopp Development Inc. announced the release of the new accessory for their latest magnetic resonance imaging (MRI) entryway system, FerrAlert Ferromagnetic Incident Log Manager (FILM). This logging device was designed specifically with The New Joint Commission standards in mind. In addition, it is a great tool to assist with internal root cause analyses, according to the company.
In their new standards Revised Requirements for Diagnostic Imaging Services, effective July 1, 2015, The Joint Commission recognizes the need for documenting ferromagnetic materials entering Zone IV (the MRI scanner room).
The Standard PI.01.01.01 of the new The Joint Commission document states:
“A 47. The [critical access] hospital collects data on the following:
● Incidents where ferromagnetic objects unintentionally entered the magnetic resonance imaging (MRI) scanner room
● Injuries resulting from the presence of ferromagnetic objects in the MRI scanner room”
To assist MRI providers with The Joint Commission compliance, Kopp Development realized that an automatic method of documenting ferromagnetic incidents is needed. Such method must allow the independent documentation of the incidents to permit corrective actions as well as root cause analyses.
The FerrAlert FILM device, which is located at the doorway of the MRI magnet room, continuously captures images. The imaging system is configured and positioned in a location to allow simultaneous viewing both inside the MRI magnet room as well as the approach to the magnet room. When a ferromagnetic object begins to pass through the FerrAlert detection portal, a signal is generated and communicated to the FILM. The FILM stores the images to preserve the history leading up to the ferromagnetic intrusion event, as well as the actions taken following the threat detection. These images are stored in an electronic folder that is date- and time-stamped.
The FerrAlert FILM device can be used only with Kopp Development’s latest entryway system, FerrAlert Halo II Plus. The Plus system dramatically reduces alarm fatigue by not alarming on the act of moving the MRI door or ferromagnetic objects exiting the MRI room. Alarm fatigue reduction is a top priority for ECRI and The Joint Commission.
For more information: www.koppdevelopment.com


 May 14, 2026
May 14, 2026