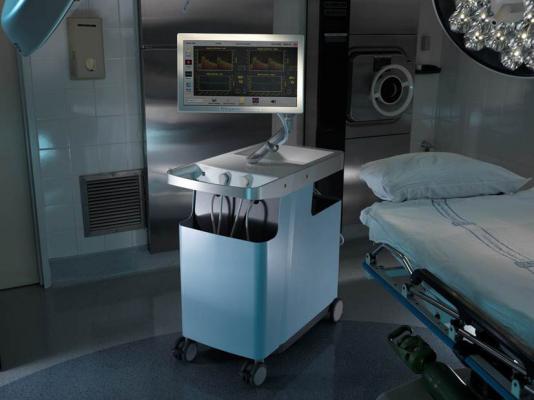
February 1, 2013 — PhysioSonics announced the U.S. Food and Drug Administration (FDA) has granted the Presto 1000 flow monitor system 510(k) clearance for cranial blood flow monitoring. In addition, the company announced completion of Series B add-on debt financing, closing with $1.05 million.
The Presto 1000 flow monitor is designed to allow any clinician to perform mid cerebral cranial blood flow monitoring, freeing institutions to more efficiently deploy sonographers who are required less often for the system. It is also designed to replace periodic measurement with continuous monitoring.
"The Presto 1000 flow monitor will help with ICU and surgical patients by detecting abnormal changes in cerebral blood flow and alerting the attending nurse, bringing further institutional resources to diagnose the cause. These flow anomalies may be indicators of severe conditions such as vasospasm from aneurysm ruptures or head trauma, disturbances with brain auto-regulation or MCA blood clots," said Brad Harlow, CEO of PhysioSonics.
PhysioSonics uses its proprietary "Flash" Doppler for the monitoring functions. The addition of Flash, with the transducers attached to headset, assists in locating and maintaining the flow of information from mid cerebral arteries.
The company has 33 U.S. and foreign patents covering its technology. It has been the recipient of DARPA and NIH funding to use PhysioSonics technology to study head trauma, traumatic brain injury and bi-lateral flow monitoring.
PhysioSonics has developed a non-invasive, long-term cerebral blood flow (CBF) Platform. Utilizing novel ultrasound technology, the company has developed a new method that enables the device to locate and hold on the cerebral artery without a trained sonographer. This auto-locating ultrasound technology thus enables an easy-to-use method for the continuous monitoring of CBF. Future uses of this platform may include emboli detection and non-invasive intracranial pressure monitoring, both of which will require additional FDA submissions.


 October 02, 2020
October 02, 2020