Aug. 8, 2012 — Vascular Solutions Inc. launched the SuperCross FT, a new flexible-tip version of its line of SuperCross microcatheters. It is designed to address the majority of complex interventional procedures in which a flexible tipped microcatheter is needed.
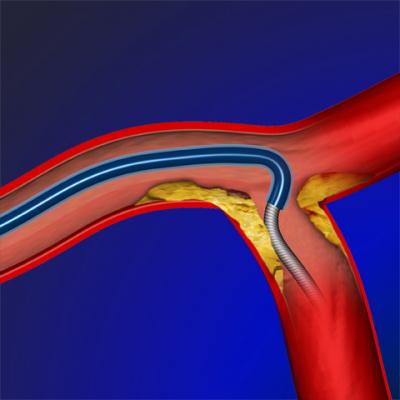
Vascular Solutions' family of SuperCross microcatheters consists of single lumen, over-the-wire microcatheters designed for guidewire support and exchange, as well as the infusion of contrast media or therapeutic agents in the coronary and peripheral vasculature. The full-length braided stainless steel construction and low profile of the SuperCross microcatheters allow access to small and tortuous vessels and enhance the ability to cross plaque lesions.
Vascular Solutions' original SuperCross microcatheter was launched in January 2011 with a straight tip that was designed for cases in which physicians needed extra support for the wire to advance past a challenging lesion. The next version introduced, SuperCross AT, features angled tip designs for directing guidewire placement in bifurcated lesions, and is offered in three preset tip configurations: 45, 90 and 120 degree angles.
"With the addition of SuperCross FT, we now have a full line of microcatheter tip designs to meet a very broad spectrum of clinical
needs," said Howard Root, CEO of Vascular Solutions. "All of our SuperCross microcatheters provide excellent deliverability and guidewire support during coronary and peripheral catheterization procedures, but we view the new FT as the new workhorse version within this product line."
Like the other models of SuperCross, the new SuperCross FT is compatible with 0.014-inch guidewires. Also like the original straight-tip model, SuperCross FT is available in 130 cm and 150 cm working lengths, has a fully embedded gold distal marker band, a clear proximal catheter hub, two depth mark indicators to aid in determining position within the guide catheter, and a hydrophilic coating on the distal 40 cm to enhance deliverability to the target vasculature.
All versions of the SuperCross microcatheters are intended to be used in conjunction with steerable guidewires to access discrete regions of the coronary and/or peripheral vasculature. They may be used to facilitate placement and exchange of guidewires and other interventional devices and to subselectively infuse/deliver diagnostic and therapeutic agents.
For more information: www.vasc.com


 September 29, 2025
September 29, 2025 


