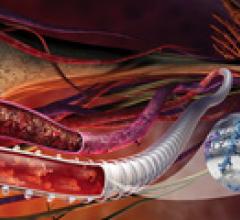
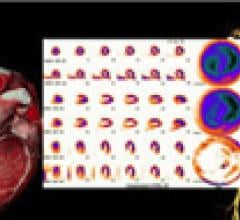
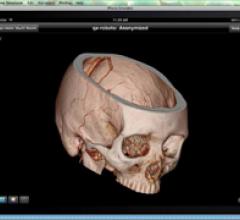
March 22, 2011 – During complex interventional procedures, it is challenging to accurately…
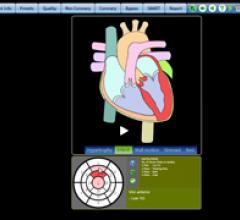
March 15, 2011 – A new graphics solution has been launched for use in professional medical…
March 15, 2011 – A new tricuspid ring for the treatment of tricuspid valve disease is approved…
March 14, 2011 – A software system that manages patient itineraries has expanded into the…
March 14, 2011 – A vascular graft for hemodialysis access has received the CE mark. The Gore…
March 11, 2011 – DR Systems will exhibit its Unity Cardiovascular Information System (CVIS) at…
March 10, 2011 – The U.S. Food and Drug Administration (FDA) has granted an abbreviated new drug…
March 10, 2011 – A newly introduced catheter provides a superior answer for percutaneous fluid…
March 8, 2011 – A new endoscopic vessel harvesting (EVH) has been launched in the United States…
March 4, 2011 – A new generation of a mobile C-arm has been launched, offering outstanding…
March 4, 2011 – The U.S. Food and Drug Administration (FDA) recently cleared two new magnetic…
March 2, 2011 – A new transseptal puncture module for education and simulation technology has…
March 1, 2011 – The U.S. Food and Drug Administration (FDA) approved azilsartan medoxomil to…
February 25, 2011 – A new upgrade program will give customers a cost-effective way to access the…
February 24, 2011 – A percutaneous microwave tissue ablation (pMTA) system will be unveiled at…
February 22, 2011 – More than 29 IHE Integration profiles were successfully certified at the IHE…
February 21, 2011 – A cardiac computed tomography (CT) report engine was approved by the U.S.…
February 18, 2011 – A zero-footprint mobile application that enables remote access to imaging…
February 18, 2011 – The U.S. Food and Drug Administration (FDA) cleared an ultrasound software…
February 16, 2011 – The newly approved drug dabigatran is an alternative to warfarin to help…

 March 22, 2011
March 22, 2011