September 3, 2015 — Results of the PLATFORM trial indicate fractional flow reserve computed tomography (FFR-CT) can obviate the need for invasive tests in up to 61 percent of patients with chest pain and suspected coronary artery disease. The results were presented at the European Society of Cardiology (ESC) Congress 2015.
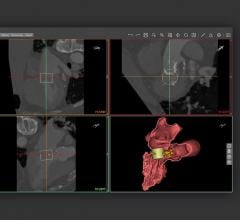
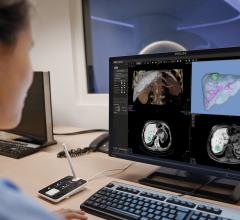
FFR-CT estimates blood flow and pressure in the coronary artery using images from computed tomography angiography (CTA).
“This approach significantly reduced the need for unnecessary invasive coronary angiography (ICA) which, although it is the gold standard for investigating chest pain, comes with the risk and costs of an invasive procedure,” said lead investigator Pamela Douglas, M.D., the Ursula Geller Professor at the Duke Clinical Research Institute, Duke University School of Medicine.
“The message is that for stable chest pain patients in whom elective ICA is recommended, consideration should be given to performing this new test first.”
When used alone, CTA can identify an abnormal narrowing (stenosis) in the coronary arteries but it cannot quantify how much this actually obstructs blood flow. Therefore “many patients who have blockages that are not interfering with blood flow may end up undergoing ICAs that show no evidence of obstructive coronary artery disease (CAD) and could possibly have been avoided, “ explained Douglas.
Other commonly used non-invasive tests for CAD, such as myocardial perfusion stress imaging or stress echocardiography, may tell whether blood supply to the heart tissue is adequate but cannot determine whether a specific blockage is obstructing blood flow, she said.
The current standard for assessing the significance of coronary artery narrowing is conventional FFR, which is invasive, involving insertion of a wire into the coronary artery.
But by using computer simulations to calculate blood flow, non-invasive FFR-CT can give a comprehensive evaluation of a patient’s chest pain without the need for an invasive procedure.
The PLATFORM study included 584 chest pain patients (average age 60.9 years) from 11 test centers in Europe.
Patients were evaluated using either FFR-CT, via the HeartFlow FFR-CT Analysis software from HeartFlow (n=297), or conventional testing (n=287) to see if FFR-CT could reduce the rate of ICAs that show no obstructive CAD.
Patients were divided into those in whom “usual care” would dictate a non-invasive investigation such as stress testing or conventional CTA (n=204) and those who would have proceeded straight to an invasive ICA (n=380).
In both groups patients were then allocated to receive either usual care (invasive or non-invasive) or FFR-CT — defined as CTA with the addition of FFR when appropriate.
Among patients already scheduled for an ICA, 73 percent in the usual care group underwent ICA only to find no significant blockages, compared to 12 percent in the FFR-CT group. Furthermore, 61 percent of patients in the FFR-CT group had the ICA cancelled based on the FFR-CT results.
While FFR-CT reduced the number of patients who underwent an ICA that found no significant disease, there was no difference between the two groups in the rate of revascularization procedures, such as stenting and coronary artery bypass surgery.
No clinical adverse events were reported at the 90-day follow-up in patients whose physicians chose to cancel ICAs based on FFR-CT guided strategy
“The study shows that CTA plus FFR-CT more effectively triages patients for invasive procedures than usual care strategies,” said Douglas.
“Although FFR-CT is a relatively new technique, PLATFORM demonstrates that it is feasible and safe with high utility in busy clinical settings.”
FFR-CT from HeartFlow received CE Mark in 2010 and received U.S. Food and Drug Administration clearance in November 2014. The technology has been evaluated in four large, clinical trials enrolling a total of more than 1,100 patients
Watch the VIDEO “Early U.S. Experience With FFR-CT in Evaluating ED Chest Pain Presentation.” A discussion with Simon Dixon, M.D., MBChB, on the use of fractional flow reserve-computed tomography (FFR-CT) to evaluate chest pain patients in the emergency department. He is chairman of the Department of Cardiovascular Medicine at Beaumont Health System and a professor of Medicine at the Oakland University William Beaumont School of Medicine. He discussed the first year of experience with FFR-CT at Beaumont Hospital in Royal Oak, Mich., during the Transcatheter Cardiovascular Therapeutics (TCT) 2016 annual meeting.
For more information: www.escardio.org

 May 12, 2020
May 12, 2020