
The European interventional cardiology market is currently valued at nearly $1.4 billion. This is a mature market that has been in decline for many years as a result of market-wide factors, such as lack of innovation, consistent price erosion and increased competition. In recent years, the interventional cardiology market has been experiencing a period of consolidation, whereby large competitors are entering new market segments organically or through strategic mergers and acquisitions. This has resulted in tremendous downward pricing pressure, as consolidated companies have more power to bundle various devices at a discount in order to win tenders and contract offers with hospitals and labs.
Recent Mergers and Acquisition in Interventional Cardiology
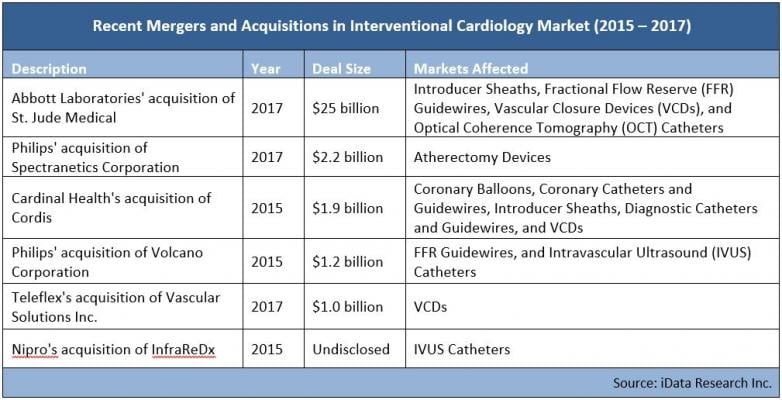
A closer analysis of recent market transactions reveals that deals are, on average, becoming increasingly large in size. This trend is not unique to interventional cardiology markets and is prevalent in other mature medical device markets as well. Market segments that are smaller in size — like vascular closure devices, optical coherenace tomography (OCT) catheters and fractional flow reserve (FFR) guidewires — will be affected the most by recent market transactions. Recent cardiology mergers include:
Philips Acquires Spectranetics, Expanding its Image-guided Therapy Device Offerings
Abbott Completes Acquisition of St. Jude Medical
Teleflex to Acquire Vascular Solutions
Philips Acquires Volcano for $1 Billion to Expand Position in Interventional Lab Market
VCD Market Has the Greatest Downside
The vascular closure devices (VCD) market segment is expected to be amongst the most negatively affected in the market, as the anticipated price erosion will outpace growth in sales volume. In addition, there has been a continual drop in the rate of complications with VCDs, mainly due to doctors becoming selective in the way that they use closure devices. This benefits physicians and patients but reduces vascular closure device sales and the market, overall.
Read the article "Advances and Trends in Vascular Closure Devices."
FFR Guidewire Market Benefiting From Price Erosion
The FFR guidewire market segment is expected to be the fastest-growing in the European interventional cardiology market, as price erosions will lead to even greater increases in device adoption and, therefore, unit sales. In addition, St. Jude Medical (now Abbott) recently released the PressureWire X Guidewire, which has demonstrated improved outcomes with complex anatomies, allowing physicians the option to use FFR guidewires in a wider range of patients. Furthermore, new companies are expected to enter this market in the coming years, which will lead to more research and development in the area.
Read the article "What is New in FFR Technology."
OCT Catheter Market
The OCT catheter market segment will also see an increase in market value, due to increased competition and adoption rates. Until recently, St. Jude Medical was the only competitor in the space and in 2017 the company was acquired by Abbott. Terumo is also gaining progress in this space with the Fastview OCT catheter. Increased competition in this market is healthy and will likely result in more research and development. In addition, adoption rates are expected to increase across Europe, due to growing awareness of the advantages of OCT, such as assisting in optimum stent placement locations and verifying correct stent deployment.
In conclusion, despite lackluster growth across the majority of the market, certain devices, such as FFR guidewires and OCT catheters, are expected to exhibit strong growth over the next several years. These markets have been affected by some major market transactions recently and will experience faster rates of price erosion, due to product bundling. However, unit sales growth will ultimately exceed price declines and lead to overall market growth for these two markets.
Editor's note: Simon Trinh is a senior research analyst at iData Research. He was the lead researcher on the latest research covering the interventional cardiology market in Europe. Jeff Wong is the analyst director at iData Research. He has been involved in numerous cardiology market research projects and now leads a team involved in that market and many others. For more information idataresearch.com.
References:


 May 13, 2026
May 13, 2026 









