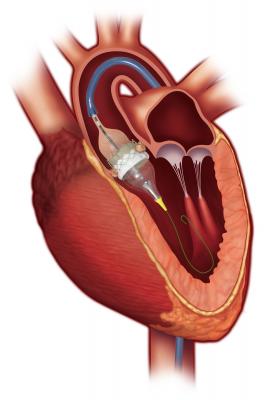
The continuation of positive clinical trial data for transcatheter aortic valve repair (TAVR), showing it is equal to or better than surgical aortic valve replacement (SAVR) was the key news from the American College of Cardiology (ACC) 2016 meeting in April. The continued positive news also made many experts at ACC begin to question the long-term future of SAVR.
TAVR is currently approved for symptomatic aortic stenosis (AS) in high-risk surgical patients and inoperable patients, as well as for failed bioprosthetic aortic valve replacement (AVR) as an alternative to conventional SAVR. Volumes have increase significantly since its U.S. Food and Drug Administration (FDA) approval, TAVR now represents as much as 30 percent of the AVR market. This percentage is expected to grow rapidly if the devices are approved for intermediate-risk surgical patients.
Aug. 18, 2016 update — FDA Expands Sapien 3 TAVR Valve Indication to Intermediate Risk Patients
Key Late-breaking Trials at ACC.16
There were three key late-breaking TAVR trials presented at ACC. The biggest news was the result of the randomized PARTNER 2 trial of the TAVR vs. SAVR in moderate-risk patients. TAVR showed similar rates of death and disabling strokes after two years compared to open-heart surgery. This study was broken into two parts because the Edwards Lifesciences Sapien XT valve was replaced part way through the trial after the approval of the improved Sapien 3 value. The results for the Sapien XT portion of the trial (PARTNER 2A) were favorable for TAVR with no difference in stroke and death at two years follow up overall. It showed superior results for TAVR in the group treated using the femoral access route. Femoral access is the preferred access in 90 percent of patients. Additionally TAVR had lower rates of major bleeding, renal failure, and atrial fibrillation and had shorter length of stay compared to SAVR.
The Sapien 3 portion of the PARTNER II trial showed the newer version of the device, which includes a skirt to reduce paravalvular leak, showed TAVR out-performed SAVR.
Watch a video interview with Vinod Thourani, M.D on the Sapien 3 beating surgery.
These results echoed the earlier results from the Medtronic CoreValve pivotal trial, which showed TAVR was superior to surgery. Three-year data from that trial presented at ACC.16 showed the divide between TAVR and SAVR continues to diverge in favor of TAVR.
Two significant concerns for TAVR remain, including the perceived higher incidence where patients require a permanent pacemaker and residual paravalvular leak, which has been associated with higher mortality. In PARTNER 2, pacemaker requirements were not different from SAVR at two years. Additionally the rate of moderate to severe aortic insufficiency (AI), or aortic regurgitation, was only 3.7 percent. Mild AI, which was more common, was not associated with adverse outcomes. These results are reassuring for TAVR.
It is highly likely that TAVR indications for lower risk patients will be approved by the FDA and reimbursed by CMS in the coming years. While there has already been creep in the commercial market into the higher end of moderate risk this will greatly open access of TAVR to a greater number of patients. Essentially all but the lowest risk patients may now be treated with TAVR and the overall market for TAVR may exceed 50 percent. Additionally the PARTNER 3 (Edwards Sapien 3) and CoreValve Low Risk (Medtronic Evolute R) trials are now enrolling patients to begin evaluating TAVR vs. SAVR in low-risk patients. Within the next five years TAVR may exceed 70 percent of the overall AVR market.
A Paradigm Shift Technology
This is disruptive technology and market forces may change how and where heart surgery is delivered in the United States. Coronary artery bypass graft (CABG) volumes have declined in the U.S. as a result of improved coronary stent technology and changes in appropriate use guidelines for revascularization. Thus far, TAVR has primarily serves an unmet need for poor surgical candidates and SAVR volumes have remained stable. However, moving forward, conventional AVR volumes are poised to decline. CABG and SAVR represent the bulk of cardiac surgical volumes at most centers. Smaller and moderate sized programs at more regional hospitals may see overall surgical volumes decline to the point that they are no longer viable. The multidisciplinary nature of TAVR also may not be feasible at many smaller programs.
Additionally with the uncoupling of percutaneous coronary interventions (PCI) in cath labs from surgical backup throughout the United States, the economics of maintaining a surgical program in smaller centers may not make sense and programs may close. Just as coronary PCI has defused from large centers, we may see cardiac surgery in general consolidating in higher volume programs. Given the clear volume outcomes relationship with TAVR, this may be good for public health. While it is hard to predict the future of this consolidation, it is a possible, if not probable, scenario driven to a large degree by the rise of TAVR.
Editor’s note: Michael J. Rinaldi, M.D., FACC, FSCAI, is the director of clinical research at Sanger Heart and Vascular Institute and a professor of medicine, CarolinasHealthCare System, Charlotte, N.C. He is an expert in TAVR, and has authored several studies that examine its applicability.


 April 24, 2026
April 24, 2026 









