January 9, 2014 — Highly invasive therapies are challenging due to the inability to quickly and safely secure devices inside defective
hearts. Sutures take too long to stitch and can cause stress on fragile heart tissue and currently available clinical adhesives are either too toxic or tend to lose their sticking power in the presence of blood or under dynamic conditions, such as in a beating heart.
"About 40,000 babies are born with congenital heart defects in the United States annually, and those that require treatment are plagued with multiple
surgeries to deliver or replace non-degradable implants that do not grow with young patients," said Jeffrey Karp, Ph.D., division of biomedical engineering, department of medicine, Brigham and Women’s Hopsital, and co-senior study author of a new study that may improve how surgeons treat congenital heart defects.
The
study, “A Blood-Resistant Surgical Glue for Minimally Invasive Repair of Vessels and Heart Defects,” appeared in
Science Translational Medicine Jan. 8.
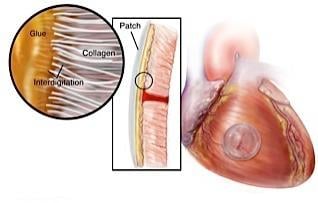
In the preclinical study, researchers from Boston Children's Hospital, BWH and Massachusetts Institute of Technology (MIT) developed a bio-inspired adhesive that could rapidly attach biodegradable patches inside a beating heart in the exact place where congenital holes in the heart occur.
Recognizing that many creatures in nature have secretions that are viscous and repel water — enabling them to attach under wet and dynamic conditions — the researchers developed a material with these properties that also is biodegradable, elastic and biocompatible. According to the study authors, the degradable patches secured with the glue remained attached even at increased heart rates and blood pressure.
"This adhesive platform addresses all of the drawbacks of previous systems in that it works in the presence of blood and moving structures," said Pedro del Nido, M.D., chief of cardiac surgery, Boston Children's Hospital, and co-senior author of the study. "It should provide the physician with a completely new, much simpler technology and a new paradigm for tissue reconstruction to improve the quality of life of patients following surgical procedures."
Unlike current surgical adhesives, this adhesive maintains sticking power in the presence of blood and in active environments.
"This study demonstrated that the adhesive was strong enough to hold tissue and patches onto the heart equivalent to suturing," said Nora Lang, M.D., department of cardiac surgery, Boston Children's Hospital, and co-first author of the study. "Also, the adhesive patch is biodegradable and biocompatible, so nothing foreign or toxic stays in the bodies of these patients."
Its adhesive abilities are activated with UV light, providing an on-demand, anti-bleeding seal within five seconds of UV light application when applied to large, high-pressure blood vessels and cardiac wall defects.
"When we attached patches coated with our adhesive to the walls of a beating heart, the patches remained despite the high pressures of blood flowing through the heart and blood vessels," said Maria Pereira, Ph.D., division of biomedical engineering, department of medicine, BWH, and co-first author of the study.
The researchers noted that their waterproof, light-activated adhesive will be useful in reducing both the invasiveness of surgical procedures and operating times, in addition to improving heart surgery outcomes.
The adhesive technology and other related platforms have been licensed to a startup company, Gecko Biomedical, based in Paris. The company has raised 8 million euros in their recently announced Series A financing round and expects to bring the adhesive to the market within three years.
The bio-inspired glue described is non-toxic, binds strongly to tissues offering a leakproof seal on demand and works well in the presence of actively contracting tissues and blood flow. The authors demonstrated how the adhesive, alone or in conjunction with a biodegradable patch, can be used to effectively repair defects (ruptures) of the heart and blood vessels during minimally invasive procedures.
The glue is based on the combination of safe, naturally occurring compounds to form a biocompatible prepolymer with tunable adhesive and mechanical properties. The glue is soft and elastic when applied to the wet surfaces of the wounds, where it adheres gently to the tissues, permitting fine adjustments or repositioning when used with the patch. The glue is activated (polymerized) upon exposure to UV light to form a strong, leakproof but flexible seal, giving the surgeon full control of the process. The adhesive can be engineered with mechanical characteristics similar to arteries and the digestive system, where initial applications are targeted. The authors also described how the composition of the prepolymer can be adjusted for strength or rate of biodegradation to suit the wound being repaired.
This research was supported by the Center for Integration of Medicine and Innovative Technology, Technology Research Program grant from Boston Children's Hospital and the National Institutes of Health (NIH) (HL73647, GM086433, DE013023).
www.geckobiomedical.com, stm.sciencemag.org