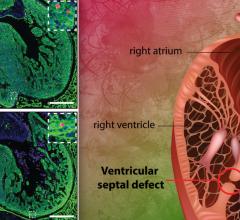
May 6, 2015 — A retrospective review of 633 adults and children who underwent bioprosthetic pulmonary valve replacement (PVR) for congenital heart disease between 1996 and 2014 indicated that the risk of re-intervention was five times greater for children than adults. In addition, the likelihood of re-intervention decreased by 10 percent for each increasing year of age at surgery. Valve type was another important determinant of re-intervention, according to Rio S. Nomoto, BA, medical student at Tufts University School of Medicine, who presented the results of this research at the 95th American Association for Thoracic Surgery (AATS) Annual Meeting in Seattle on April 28.
PVR is recommended for both asymptomatic and symptomatic patients with CHD who have increased risk for right ventricular dilation and dysfunction. These patients may exhibit exercise intolerance, arrhythmia and sudden cardiac events. Pulmonary regurgitation is a common indication for PVR.
“In this comprehensive series, we have identified specific valve type and all measures related to younger patient age at surgery to be important predictors of re-intervention,” explained lead investigator Christopher W. Baird, M.D., of the Department of Cardiac Surgery at Boston Children’s Hospital of Harvard Medical School. The researchers also found that patients who were small for their age were much more likely to experience valve failure, suggesting that these children merit more intensive long-term follow-up.
In this group of 633 patients, the median age at surgery was 17.5 years, and 49 percent of the patients were younger than 18 years. Eight patients were infants. Sixty-eight percent of cases had a diagnosis of Tetralogy of Fallot. The median follow-up was 2.9 years.
Six percent of patients (44) required re-intervention. Of these, 17 underwent a surgical re-intervention and 27 had cardiac catheterization to replace the pulmonary valve. Most events precipitating re-intervention happened at least three years after initial surgery. Thirty patients required more than one re-intervention and 19 deaths occurred post-operatively or during follow-up.
Age at surgery was an important predictor of future re-intervention. At five years post-op, the re-intervention rate was 11 percent for patients younger than 12 years, 7 percent for those 12-27 years old and 0 percent for those older than 27 years. At 10 years after surgery, 100 percent of children who had been operated on when they were less than 6 years old required repeat surgery, as did 74 percent of those who had been between 6 and 11 years old; in contrast, the re-intervention rate was 22 percent for those age 12 to 17 years and 10 percent for those between 18 and 26 years old. None of the patients who were 27 years or older at the time of surgery required re-intervention at the 10-year mark, noted Nomoto.
Other factors associated with younger age were also found to be risk factors for re-intervention, such as low body weight, small body surface area and body mass index (BMI). For instance, those younger than 20 years of age with BMI more than two standard deviations below normal were seven times more likely to experience valve failure. “Children with low BMI may merit closer monitoring following PVR or nutritional counseling to increase BMI prior to scheduling surgery,” suggested Baird.
There was also a significant difference in the risk of re-intervention according to valve type. Fifty percent of patients received the Sorin Mitroflow, 35 percent the CE Magna or MagnaEase, 11 percent the CE Perimount and 4 percent porcine values. Of the four valves evaluated, the Sorin Mitroflow exhibited a significantly higher re-intervention rate (17.6 percent) than the others.
Neither larger valve size nor BSA-adjusted valve orifice size predicted re-intervention. The investigators also found that the magnitude of the valve type differences was independent of age.
For more information: www.aats.org


 March 31, 2025
March 31, 2025