Photo: Getty Images
April 24, 2026 — A new analysis from the NOAH – AFNET 6 trial shows that artificial intelligence (AI) can help unlock data from pacemakers to better understand stroke risk in patients with device-detected atrial fibrillation (AF). The study found that patients who spent more time in AF did not have a higher risk of stroke or other cardiovascular events than those who spent less time in AF. Researchers presented these findings today as a late-breaking clinical trial at Heart Rhythm 2026.
As AF prevalence continues to increase worldwide, clinicians face growing pressure to move beyond simply detecting the presence of AF, and toward understanding its clinical impact. AF burden — the amount of time a patient spends in AF —has emerged as an important factor linked to stroke risk and outcomes, yet it remains difficult to capture at scale in clinical research and routine practice.
NOAH–AFNET 6 (Non-vitamin K antagonist Oral anticoagulants in patients with Atrial High-rate episodes) is an investigator-initiated trial evaluating anticoagulation in patients with device-detected AF. The main trial found that anticoagulants slightly reduced stroke risk but increased major bleeding, leading to early termination due to an unfavorable balance between benefits and risks.1-9
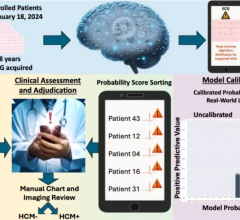
This substudy of NOAH-AFNET 6 shows that AI-driven models using natural language processing can automatically and reliably extract atrial fibrillation (AF) burden information from routine pacemaker reports, supporting more individualized risk assessment and potentially helping clinicians make more informed decisions about therapies such as anticoagulation. Researchers applied this approach within NOAH–AFNET 6, analyzing 11,964 pacemaker reports from 2,534 patients with device-detected AF to better understand how AF burden relates to clinical outcomes and response to the anticoagulant edoxaban.
“With atrial fibrillation on the rise worldwide, we need better ways to use existing clinical data to understand not only whether patients have AF, but how much of it they experience over time,” says Ulrich Schotten, MD, PhD, Professor of Cardiac Electrophysiology at Maastricht University in the Netherlands. “This AI-driven large language model approach allows us to unlock meaningful insights from data we already collect and use them to advance research and improve patient care.”
Investigators found that the model identified AF burden, or “mode switch burden,” in more than 70% of reports, showing strong applicability to real-world data. In a validation subcohort, the large language model matched manual review in more than 98% of cases when AF burden data were available, and AF burden and mode switch burden showed high concordance, supporting the reliability of device-based measures. Baseline AF burden remained low across the study population. Over a median follow-up of 19 months, the magnitude of AF burden did not relate to thromboembolic risk, with low stroke rates even among patients with higher AF burden, and AF burden also did not modify the effect of anticoagulation therapy. Anticoagulation increased the risks of bleeding and death, regardless of AF burden.
“These results highlight the need to better understand the effects of low-burden AF on stroke risk,” says Paulus Kirchhof, MD, Director of the Department of Cardiology at the University Heart and Vascular Center UKE Hamburg. “This analysis provides a clear signal: low AF burden is associated with low stroke risk. Larger datasets will be needed to define which levels of AF burden warrant anticoagulation and which may instead require rhythm control strategies.”
References
-
Kirchhof P, Toennis T, Goette A, et al. Anticoagulation with Edoxaban in Patients with Atrial High-Rate Episodes. N Engl J Med 2023; 389:1167-1179. DOI:10.1056/NEJMoa2303062.
-
Toennis T, Bertaglia E, Brandes A, et al. The influence of Atrial High Rate Episodes on Stroke and Cardiovascular Death - An update. Europace. 2023 Jul 4;25(7). DOI:10.1093/europace/euad166.
-
Becher N, Toennis T, Bertaglia E, et al. Anticoagulation with edoxaban in patients with long Atrial High-Rate Episodes ≥24 hours. Eur Heart J. 2024 Mar 7;45(10):837-849. DOI:10.1093/eurheartj/ehad771
-
Lip YH, Nikorowitsch J, Sehner S et al. Oral anticoagulation in device-detected atrial fibrillation: effects of age, sex, cardiovascular comorbidities, and kidney function on outcomes in the NOAH-AFNET 6 trial. Eur Heart J. 2024 April 9. DOI:10.1093/eurheartj/ehae225
-
Diener HC, Becher N, Sehner S, Toennis T et al. Anticoagulation in patients with device-detected atrial fibrillation with and without a prior stroke or transient ischemic attack. The NOAH-AFNET 6 trial. J Am Heart Assoc. 2024 Sep 3;13(17):e036429. DOI:10.1161/JAHA.124.036429
-
McIntyre WF, Benz AP, Becher N, et al. Direct Oral Anticoagulants for Stroke Prevention in Patients with Device-Detected Atrial Fibrillation: A Study-Level Meta-Analysis of the NOAH-AFNET 6 and ARTESiA Trials. Circulation. 2024 Mar 26;149(13):981-988. DOI:10.1161/CIRCULATIONAHA.123.067512
-
Schnabel RB, Benezet-Mazuecos J, Becher N, McIntyre WF et al. Anticoagulation in patients with device-detected atrial fibrillation with and without concomitant vascular disease – A combined secondary analysis of the NOAH-AFNET 6 and ARTESiA trials. Eur Heart J. 2024 Dec 7;45(46):4902-4916. DOI:10.1093/eurheartj/ehae596
-
Becher N, Koellner G, Blomstrom-Lundqvist C, Camm AJ et al. Effects of anticoagulation in patients with device-detected atrial fibrillation and multiple stroke risk factors: A Win Ratio analysis of the NOAH-AFNET 6 trial. Eur Heart J Qual Care Clin Outcomes. 2025 Dec 19;11(8):1351-1358. DOI:10.1093/ehjqcco/qcaf087
-
Goette A, Lemoine MD, Fierenz A et al. Kidney disease increases the risk of cardiovascular events in patients with device-detected atrial fibrillation: NOAH-AFNET 6. Europace. 2026. DOI:10.1093/europace/euag083
Session Details:
“Late-Breaking Clinical Trials: Clinical Electrophysiology: Distribution of atrial fibrillation burden, relation to outcomes, and effectiveness and safety of anticoagulation based on device reports: The NOAH-AFNET 6 trial” [Friday, April 24, 2026, at 10:45 am CT]


 May 20, 2026
May 20, 2026