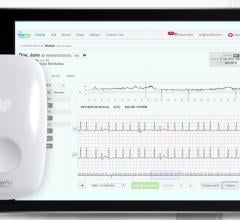
March 27, 2015 — AliveCor Inc. announced the launch of a cloud-based electrocardiogram (ECG) analytics program with Preventice Solutions. Using AliveCor’s proprietary algorithms in this pilot program, Preventice Solutions intends to improve its internal workflow by reading the most critical ECGs first. This is a limited pilot program where all ECGs have their final interpretation provided by the independent diagnostic testing facility of eCardio, which is owned by Preventice Solutions; it is the beginning of a new platform for AliveCor’s proprietary algorithms.
AliveCor is known for its AliveCor Heart Monitor and its AliveECG mobile app that uses U.S. Food and Drug Administration (FDA)-cleared algorithms to analyze ECGs for potential arrhythmias. For the first time, the company will use their algorithms to analyze ECGs from traditional cardiac event monitors as well. This potential expansion of AliveCor’s services may extend its automated ECG analytics to cardiac monitoring companies like Preventice Solutions and hospital cardiology departments, providing an easy to use, physician-led way of monitoring patients for suspected or diagnosed arrhythmias.
This strategic program expands upon AliveCor and Preventice Solutions’ current relationship. Through the AliveECG app, AliveCor users are provided with an analysis service that offers expert review of ECGs, powered by eCardio independent monitoring diagnostic facility owned by Preventice Solutions. Users can send ECG readings to remote cardiac technicians for detailed interpretation, at any time, providing users with a more complete picture of their current heart health. Patients can either pay $2 to get their report back in 24 hours, or $5 to get their report within 30 minutes.
For more information: www.alivecor.com


 July 22, 2025
July 22, 2025