October 28, 2013 — Alivecor Inc. announced that the AliveCor Heart Monitor is now being offered to participants in the Health eHeart Study with 2,000 units to be deployed before the end of the year. The University of California, San Francisco’s (UCSF) Health eHeart Study aims to enroll one million participants and monitor heart health for at least 10 years.
The goal of the study is to better understand which factors impact heart health long-term and thereby develop better methods to predict, prevent and treat cardiovascular disease.
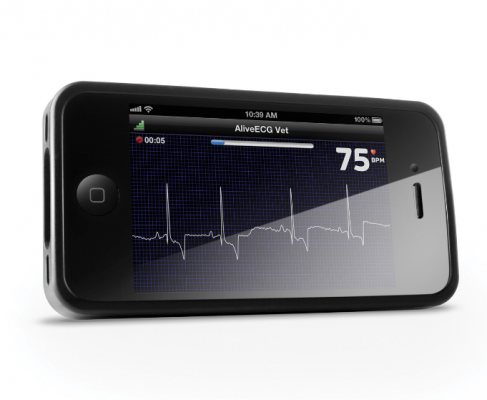
The AliveCor Heart Monitor is the only mobile
electrocardiogram (ECG) device that supports both iPhone and Android compatible
smartphones. It records, displays, annotates, stores and transfers single-channel ECG rhythms wirelessly using the free AliveECG application.
Overall ease of use and convenience is further expanded with AliveInsights for tracking of heart rate and the Enhanced Filter that improves visual output of the ECG. Over 300,000 ECGs have been recorded to date with the heart monitor. With secure storage in the Cloud, patients can access their data confidentially any time, anywhere and can grant access to their physician.
“The goal of Health eHeart Study is to expand on traditional research studies, which have identified common risk factors for heart disease by studying several thousand people with yearly or biannual visits to the clinic for tests and examination, such as an ECG,” said Jeffrey Olgin, M.D., chief of the division of cardiology, UCSF and one of the principal investigators, Health eHeart Study. “The Health eHeart Study expands on these types of studies by being able to track participants anywhere in the world and receive data any time they choose to send their information. The AliveCor Heart Monitor helps us collect ECG data on large numbers of people and at much more frequent intervals simply through their smartphone.”
The AliveCor Heart Monitor is available for purchase in the United States to medical professionals and patients with prescriptions.
For more information: www.alivecor.com