
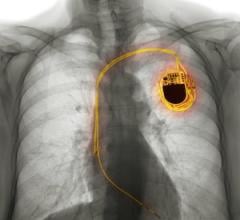
May 21, 2015 — Biotronik announced that since the launch of the ProMRI Eluna pacemaker system in late March 2015, 60 percent of pacing devices sold by the company are approved for use during magnetic resonance imaging (MRI) scans. The announcement was made at Heart Rhythm 2015, the Heart Rhythm Society’s 36th annual scientific sessions, May 13-16 in Boston.
“The majority of pacemaker patients are over the age of 65 and most are likely to have a clinical need for an MRI scan after their device is implanted,” said J. Rod Gimbel, M.D., cardiac electrophysiologist at Cardiology Associates of East Tennessee. “The pacemaker patient population typically has multiple orthopedic, oncologic, neurologic and cardiovascular co-morbidities that are best understood with MRI; thus, it is crucial that we implant pacemakers that allow continued unfettered access to MRI.
“In my practice, I implant MRI conditional devices and leads at every opportunity to do so, as I believe MRI conditional devices are the standard of care,” added Gimbel.
More than 6 million people aged 65 or older undergo an MRI scan in the United States every year and 20 percent of pacemaker patients need an MRI within the first two years of implant. According to the Mayo Clinic, approximately 75 percent of patients who have an implantable cardiac device will need an MRI in their lifetime.
Biotronik’s ProMRI technology allows patients to undergo full-body MRI scans with both single-chamber (SR-T) and dual-chamber (DR-T) pacemakers when implanted with specially designed and thoroughly tested Biotronik pacing leads.
For more information: www.biotronik.com

 April 24, 2026
April 24, 2026 









