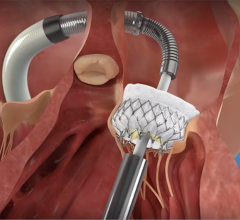
January 5, 2024 — Medtronic, a global leader in healthcare technology, today announced it has received CE (Conformité Européenne) Mark for its Micra AV2 and Micra VR2, the next generation of its industry-leading miniature, leadless pacemakers.
Micra AV2 and Micra VR2, the world's smallest pacemakers, provide longer battery life and easier programming than prior Micra pacemakers, while still delivering the many benefits of leadless pacing such as reduced complications compared to traditional pacemakers.2
With approximately 40% more battery life compared to previous generations,1 Medtronic projects the battery life of Micra AV2 and Micra VR2 is nearly 16 and 17 years, respectively.3 This means more than 80% of patients who receive a Micra may only require one device for life.3
This CE Mark milestone follows U.S. Food and Drug Administration approval for the Micra AV2 and VR2 devices in 2023.
In addition to size and longevity benefits, Micra devices are also the only leadless pacemakers with remote monitoring capabilities, which allow a doctor or clinic to check on a patient's heart device without the need for a patient to travel to their clinic for an in-person appointment. Other patient benefits of remote monitoring may include a reduction in hospitalizations and increase in sense of security.4,5
"For more than eight years, our Micra leadless pacemakers have provided meaningful benefits to people in Europe who require a pacemaker. Now, these patients have access to the latest leadless pacing technology that, for most of them, may be the only device they will ever need," said Robert C. Kowal, M.D., Ph.D., general manager, Cardiac Pacing Therapies within the Cardiac Rhythm Management business, which is part of the Cardiovascular Portfolio at Medtronic.
"The first generation of leadless Micra pacemakers demonstrated a significant reduction in major complications, and this next generation of Micra brings several additional benefits including greater longevity, and specifically for the Micra AV2, new algorithms to optimize AV synchrony at faster heart rates while requiring less in-office programming," said Prof. Dr. Christophe Garweg, Cardiologist at University Hospitals of Leuven, Belgium. "These advantages likely will expand the use of the leadless pacemaker to more patients."
Globally, more than 200,000 patients have received Micra pacemakers.6 Comparable in size to a large vitamin, Micra pacemakers are less than one-tenth the size of traditional pacemakers. Unlike traditional pacemakers, Micra pacemakers do not require leads or a surgical "pocket" under the skin, so potential sources of complications related to leads and pockets are eliminated, and there is no visible sign of the device.
Leaders in Cardiac Pacing
Receiving CE Mark for Micra AV2 and VR2 builds on the Medtronic legacy in cardiac pacing, which includes:
- Inventing the first wearable pacemaker in 1957.
- Launching the first pacemaker for use in MRIs in 2011.
- Creating and launching the first leadless pacemaker in 2015.
- Launching the first leadless pacemaker to coordinate the heart's electrical signals by sensing atrial activity, without a lead or device in the atria, in 2020.
- Gaining regulatory approval for the first pacing system to activate the heart's natural electrical system, conduction system pacing (approval via the His-bundle in 2018 and via the left bundle branch area in 2022).
- Receiving U.S. approval for Micra AV2 and VR2 in 2023.
For more information: www.medtronic.com
References:
1 Escalante K. Micra AV2 and Micra VR2 longevity comparison. 2023. Medtronic Data on File.
2 El-Chami MF, Al-Samadi F, Clementy N, et al. Updated performance of the Micra transcatheter pacemaker in the real-world setting: A comparison to the investigational study and a transvenous historical control. Heart Rhythm. December 2018;15(12):1800-1807.
3 Sheldon T, Escalante K, and Fagan D. Device Longevity and AV Synchrony Algorithm Modeling of a Leadless Pacemaker Family: A Virtual Patient Analysis. January 2023. Medtronic Data on File.
4 Piccini JP, Mittal S, Snell J, Prillinger JB, Dalal N, Varma N. Impact of remote monitoring on clinical events and associated health care utilization: A nationwide assessment. Heart Rhythm. 2016 Dec;13(12):2279-2286. doi: 10.1016/j.hrthm.2016.08.024. Epub 2016 Aug 17. PMID: 27544748.
5 Artico J, Zecchin M, Fantasia AZ, Skerl G, Ortis B, Franco S, Albani S, Barbati G, Cristallini J, Sinagra G. Long-term patient satisfaction with implanted device remote monitoring: a comparison among different systems. Journal of Cardiovascular Medicine. 2019 Aug 1;20(8):542-50.
6 Leick, A. Micra "Largest Reach" supporting data. September 2023. Medtronic Data on file.


 July 08, 2024
July 08, 2024