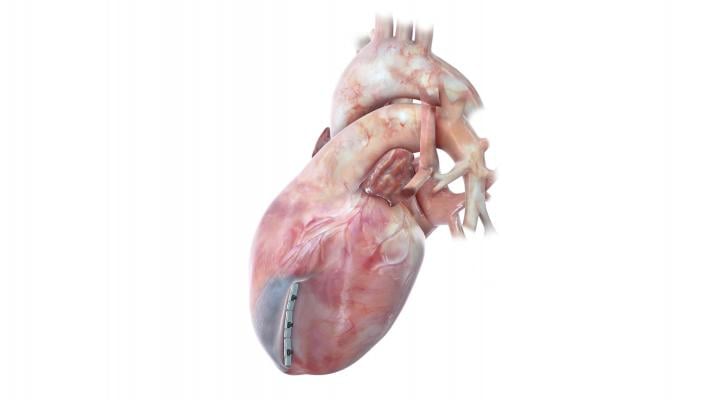
December 18, 2019 — BioVentrix, Inc., developer of the first less invasive system for left ventricular remodeling, today announced online publication of positive one-year results from its CE Mark study of the Revivent TC System in the European Journal of Heart Failure. The paper will be published in print in the January issue of the journal.
Authors of the study, entitled “Less Invasive Ventricular Reconstruction for Ischemic Heart Failure” concluded, “Treatment with the Revivent TC System in patients with symptomatic heart failure results in significant and sustained reduction of left ventricular (LV) volumes and improvement of LV function, symptoms, and quality of life.”
They added, “The ability to achieve these results without the need for sternotomy or cardiopulmonary bypass is an important advance for the treatment of patients suffering from ischemic cardiomyopathy heart failure.”
The prospective, multicenter, single-arm study of 89 patients at 22 centers in 12 countries in the European Union was designed to evaluate the efficacy and safety of the Revivent TC System for scar exclusion in the heart, volume reduction, and reshaping of the LV in selected patients with ischemic cardiomyopathy (enlarged and weakened left ventricle with reduced capacity to pump blood after a heart attack). Of the 86 patients successfully treated with the system, 51 patients received delivery via a sternotomy and 35 were treated using a less invasive, hybrid approach without sternotomy. None of the patients treated in this trial required cardiopulmonary bypass to implant the device.
The study showed the following patient outcomes at 12 months:
- 100 percent of patients demonstrated significant and sustained reduction in LV volumes
- 27 percent reduction in LVESVI (74 ± 28 ml/m²BSA vs 54 ± 23 ml/m²BSA, P<0.001)
- 24 percent reduction in LVEDVI (106 ± 33 ml/m²BSA vs 80 ± 26 ml/m²BSA, P<0.0001)
- 16 percent improvement in LVEF (29 ± 8% vs 34 ± 9%, P<0.005)
- 90.6 percent survival
- 27 percent improvement in New York Heart Association (NYHA) functional class (average class of 2.6 vs. 1.9) – 59% of subjects were NYHA class III at baseline; at one year, 79.4% were class I or II
- 36 percent improvement in quality of life as measured by the Minnesota Living with Heart Failure questionnaire (MLHF) (41 vs. 27 points, p<0.001)
- 18 percent improvement in exercise capacity as measured by the Six Minute Walk Test (6MWT – 342 meters vs. 403 meters, p<0.001))
- 50 percent improvement in average mitral regurgitation (MR) grade in the 68 patients who entered study with measurable functional MR (FMR) of at least grade 1+ at six months (1.12 vs. 0.57) and 24 percent improvement at 12 months (1.12 vs. 0.86)
- 97 percent procedural success
- Median length of hospital stay was 14 days; however, subsequent post-market study has seen length of stay decrease substantially as implanters gain more experience
Percutaneous coronary intervention (PCI), which implants a stent, is considered the standard of care for restoring blood flow to the heart after a heart attack. However, 50 percent of patients treated with PCI still develop large LV scars, which cause the heart to dilate. Enlarged LVs are associated with more severe heart failure symptoms, higher mortality and more frequent re-hospitalization. Positive remodeling of the heart has been shown to improve LV function and heart failure symptoms. The only currently available option is surgical ventricular remodeling, which is highly invasive.
The company is currently enrolling patients in the REVIVE-HF post-market study in Europe, a randomized, controlled, prospective, multi-center, dual-arm study evaluating the Revivent TC system against Guideline-Directed Medical Therapy. The study is expected to enroll 180 patients.
In the U.S., the company is currently enrolling patients in a pivotal trial of the Revivent TC system, the ALIVE Trial. View a testimonial from a U.S. patient who has received the Revivent TC system here.
For more information: www.bioventrix.com


 May 13, 2026
May 13, 2026 









