July 12, 2018 — CathWorks announced the approval of a new Current Procedural Terminology (CPT) code 0523T for non-invasive, 3-D FFRangio-enabled interpretation of possible atherosclerotic stenosis during coronary angiography interventions. The company said the code issuance is a major step forward in helping physicians objectively and cost-effectively determine if percutaneous coronary intervention (PCI) is indicated and revascularization has occurred in every coronary angiography procedure.
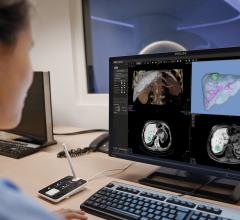
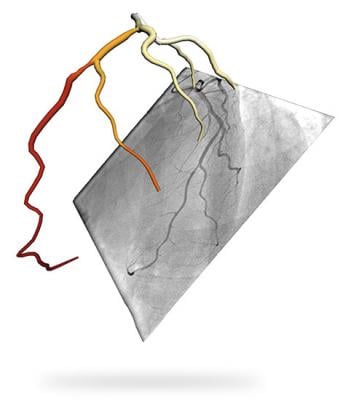
The CathWorks FFRangio System allows objective intraprocedural decision-making. The non-invasive fractional flow reserve (FFR) platform quickly and precisely delivers objective multi-vessel physiologic measurements to cost-effectively optimize and confirm intraprocedural PCI therapy decisions.
CPT code 0523T enables healthcare providers using the system to be reimbursed for:
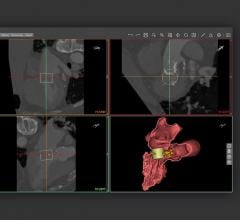
Intraprocedural coronary fractional flow reserve (FFR) with 3-D functional mapping of color-coded FFR values for the entire coronary tree, derived from coronary angiogram data that allow real-time review and interpretation of possible atherosclerotic stenosis(es) intervention.
The CathWorks FFRangio System is in development and is not yet U.S. Food and Drug Administration (FDA)-cleared.
For more information: www.cath.works
Related FFRangio Content
VIDEO: Angiography Image-based FFR May Eliminate Need for Pressure Wires

 May 12, 2020
May 12, 2020