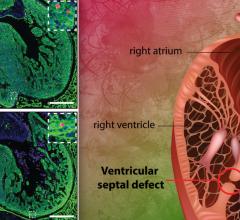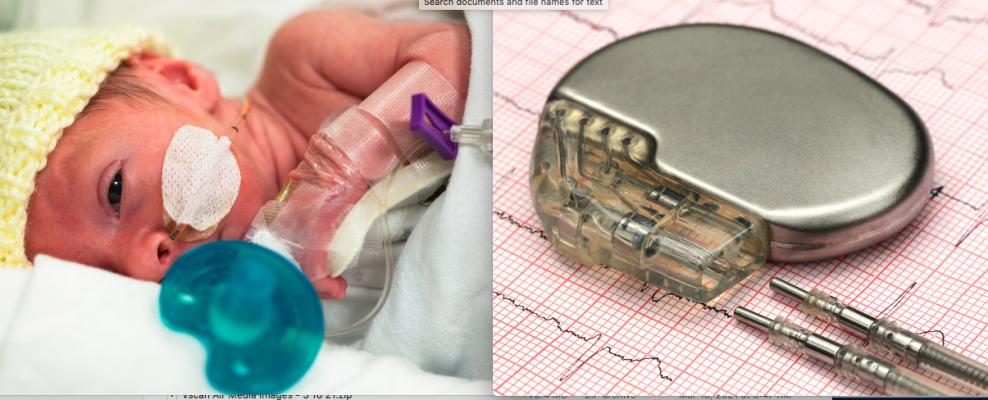
March 24, 2021 — Congenital heart disease (CHD) affects six out of 1,000 babies born in the U.S. each year and is often complicated by arrhythmias. While the last decade brought great advances in technologies that improve the care of adult arrhythmias, pediatric patients have been left behind, with only five devices approved for use in children in the same period. As a result, pediatric specialists are often using off-label or improvised electrophysiology (EP) devices to treat pediatric arrhythmias, including in the smallest newborns.
Recognizing this unmet need, the National Capital Consortium for Pediatric Device Innovation (NCC-PDI), in collaboration with MedTech Innovator, is accepting applications through April 12, 2021, for its annual "Make Your Medical Device Pitch for Kids!" competition. This year's competition focuses on innovations in pediatric devices that treat CHD, with an emphasis on electrophysiology devices such as pacemaker systems, ablation catheters, wearable monitoring devices and related technologies that address arrhythmias in children.
"NCC-PDI was created, with the support of the Food and Drug Administration (FDA), to seek out and address significant unmet needs in pediatric medical devices," saaid Kolaleh Eskandanian, Ph.D., M.B.A., P.M.P., vice president and chief innovation officer at Children's National Hospital and principal investigator of NCC-PDI. "We have learned from the experts that pediatric-specific technologies for treating arrhythmias would be a game changer in the care of their patients, so we are focusing our competition and grant awards on this opportunity."
Using a virtual format, semi-finalists chosen from all submissions will make their first pitch on May 12, 2021. Up to 10 finalists selected from this event earn participation in a special pediatric-focused track of the MedTech Innovator accelerator program, the largest medtech accelerator in the world, beginning in June 2021. These innovators then participate in the pediatric competition finals in September 2021 where judges will award up to $150,000 in FDA-sponsored grants to the devices selected as most impactful and commercially viable.
"Because pediatric medical devices face unique challenges in achieving commercialization, MedTech Innovator's accelerator program provides specialized mentoring and guidance from experienced industry leaders," said Paul Grand, CEO of MedTech Innovator. "Our program is designed to provide immediate and long-term benefits to help innovators more rapidly achieve success on their journey to improving health care for children."
How significant is the need for pediatric devices to address arrhythmias? In a recent survey of members conducted by the Pediatric and Congenital Electrophysiology Society (PACES), the vast majority (96%) said they believe there is a deficiency in devices available to serve the needs of pediatric patients. Conducted with the U.S. FDA, the survey also asked respondents to identify the biggest unmet need, which physicians identified as cardiovascular implantable electronic devices that are smaller, have better battery life and have pediatric-specific algorithms. Specifically, a leadless pacemaker designed for pediatric care was consistently on the most-wanted list.
"It's evident that technologies for pediatric patients with CHD are not adequate and the experts are telling us what they need," said William E. Bentley, Ph.D., Robert E. Fischell distinguished professor and director of the Robert E. Fischell Institute for Biomedical Devices at the University of Maryland. "I look forward to seeing the critical innovations that NCC-PDI will help to advance through this competition."
NCC-PDI is one of five members in the FDA's Pediatric Device Consortia Grant Program created to support the development and commercialization of medical devices for children, which lags significantly behind the advancement of adult medical devices. NCC-PDI is led by the Sheikh Zayed Institute for Pediatric Surgical Innovation at Children's National Hospital and the A. James Clark School of Engineering at the University of Maryland with support from partners MedTech Innovator, BioHealth Innovation and design firm Archimedic.
Eskandanian said that enhancing access to resources for pediatric innovators is also one of the aims of the Children's National Research and Innovation Campus, a first-of-its-kind focused on pediatric health care innovation, with the first phase currently open on the former Walter Reed Army Medical Center campus in Washington, D.C. With its proximity to federal research institutions and agencies, universities, academic research centers, as well as on-site incubator Johnson & Johnson Innovation – JLABS, the campus provides a rich ecosystem of public and private partners which, like the NCC-PDI network, will help bolster pediatric innovation and commercialization.
For more information and to apply for the upcoming NCC-PDI pitch competition: https://innovate4kids.smapply.io/prog/2021/


 March 31, 2025
March 31, 2025