Nov. 5, 2014 — Enrollment is open for the Dual Epicardial and Endocardial Procedure (DEEP) clinical trial following U.S Food and Drug Administration Approval (FDA). The DEEP trial is a prospective, multicenter, single arm, pivotal study to evaluate patients with persistent or long-standing persistent atrial fibrillation. This is the first pivotal study of its kind in the United States.
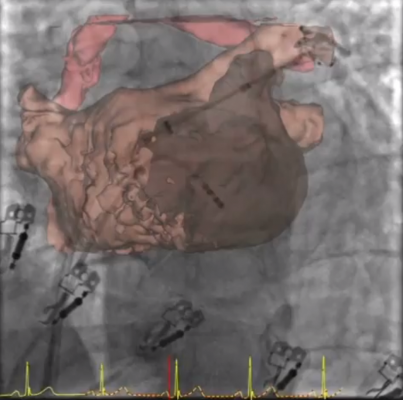
The study is designed to evaluate the safety and efficacy of the DEEP approach in treating non-paroxysmal fibrillation. The DEEP approach utilizes the specialized skills of both the cardiac surgeon and electrophysiologist (EP) to treat more severe cases of Afib, such as non-paroxysmal, which historically have been the most difficult to treat.
In the DEEP study, the cardiac surgeon and EP work together to perform a minimally invasive epicardial ablation and endocardial catheter-based ablation. The primary effectiveness endpoint is freedom from Afib and freedom of Class I or III antiarrhythmic drug therapy. AtriCure has FDA approval to enroll 220 subjects at up to 25 sites in the U.S. and internationally.
The principal investigators for DEEP are Kenneth Ellenbogen, M.D., Chairman of the Division of Cardiology at Virginia Commonwealth University (VCU) Pauley Heart Center; Dr. Paul Wang, Director, Stanford Arrhythmia Service, Professor of Medicine and Bioengineering, by courtesy, Stanford University School of Medicine; Vigneshwar Kasirajan, M.D., Chairman, Department of Surgery and Division of Cardiothoracic Surgery and Professor, Department of Surgery at VCU Pauley Heart Center; and Ali Khoynezhad, M.D., Professor of Cardiovascular Surgery at Cedars-Sinai Heart Institute.
Atrial fibrillation is the most common type of arrhythmia and can cause blood clots, stroke, heart failure and other heart–related complications. A patient with Afib has a high risk of stroke because the irregular heartbeat allows blood to pool in the left atrial appendage (LAA) and form clots that can travel through the body and into the brain.
Atrial fibrillation patients who have failed antiarrhythmic drug therapy and received up to two failed catheter ablations are candidates for the DEEP study, which is performed in two phases. In the first phase, the surgeon will make small incisions to perform endoscopic epicardial ablation using the AtriCure Bipolar System, and occlude the LAA with the AtriClip LAA Exclusion System. Then about three months later, the patient will undergo an endocardial mapping and catheter ablation procedure performed by the EP.
For more information: www.atricure.com


 May 06, 2026
May 06, 2026 









