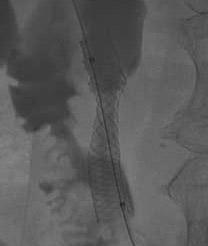
August 31, 2009 – IDEV Technologies Inc. announced last week the first patient was enrolled in the company's FDA-approved multicenter clinical trial of its self-expanding SUPERA stent for the treatment of biliary and peripheral artery disease (PAD) in the superficial femoral artery (SFA).
The clinical trial, called SUPERB (Comparison of the SUPERA PERipheral System to a Performance Goal Derived from Balloon Angioplasty Clinical Trials in the Superficial Femoral Artery), is a prospective, single-arm trial of 258 patients at up to 40 U.S. sites. It is led by national coprincipal investigators Kenneth Rosenfield, M.D., section head for vascular medicine and intervention at Massachusetts General Hospital, Boston, and Lawrence A. Garcia, M.D. chief, section interventional cardiology and associate director of vascular medicine at Boston’s Caritas St. Elizabeth Medical Center, where the first patient was treated.
The objective of the trial is to demonstrate the safety and effectiveness of the SUPERA stent in the treatment of obstructive arterial disease in the superficial femoral artery of the lower extremity. The SUPERA Interwoven Self-Expanding Nitinol Stent Transhepatic Biliary system received 510(k) clearance for palliative treatment of biliary strictures produced by malignant neoplasms and the SUPERA Interwoven Self-Expanding Nitinol Stent System received CE mark approval in Europe for biliary and peripheral vascular indications.
"The SUPERA stent has unique properties that may be demonstrated on patients at facilities throughout the U.S.," Dr. Rosenfield said. "We believe this could prove to offer a significant advance in treating PAD and helping physicians treat the most difficult of blockages in a very large underserved patient population."
"Ultimately, through this study, the SUPERA stent could become the new benchmark for stenting infra-inguinal femoral arterial obstructive disease," Dr. Garcia added.
Peter Soukas, M.D., director of vascular medicine and the vascular laboratories at St. Elizabeth's, enrolled and treated the trial's first patient.
"Our patient had a calcified SFA lesion that was treated thanks to the radial strength and flexibility afforded by the unique woven design of the SUPERA stent," Dr. Soukas said. "I am confident the stent will demonstrate the same resistance to strut fracture and primary patency that has been seen in Europe."
For more information: www.idevmd.com


 November 24, 2025
November 24, 2025 









